IL-18R supported CAR T cells targeting oncofetal tenascin C for the immunotherapy of pediatric sarcoma and brain tumors
- PMID: 39572158
- PMCID: PMC11580246
- DOI: 10.1136/jitc-2024-009743
IL-18R supported CAR T cells targeting oncofetal tenascin C for the immunotherapy of pediatric sarcoma and brain tumors
Abstract
Background: Oncofetal splice variants of extracellular matrix (ECM) proteins present a unique group of target antigens for the immunotherapy of pediatric cancers. However, limited data is available if these splice variants can be targeted with T cells expressing chimeric antigen receptors (CARs).
Methods: To determine the expression of the oncofetal version of tenascin C (TNC) encoding the C domain (C.TNC) in pediatric brain and solid tumors, we used quantitative reverse transcription PCR and immunohistochemistry. Genetically modified T cells were generated from human peripheral blood mononuclear cells and evaluated in vitro and in vivo.
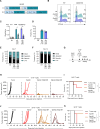
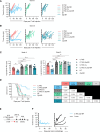
Results: We demonstrate that C.TNC is expressed on a protein level in pediatric tumors, including diffuse intrinsic pontine glioma, osteosarcoma, rhabdomyosarcoma, and Ewing sarcoma. We generate C.TNC-CAR T cells and establish that these recognize and kill C.TNC-positive tumor cells. However, their antitumor activity in vivo is limited. To improve the effector function of C.TNC-CAR T cells, we design a leucine zipper-based chimeric cytokine receptor that activates interleukin-18 signaling pathways (Zip18R). Expression of Zip18R in C.TNC-CAR T cells improves their ability to secrete cytokines and expand in repeat stimulation assays. C.TNC-CAR.Zip18R T cells also have significantly greater antitumor activity in vivo compared with unmodified C.TNC-CAR T cells.
Conclusions: Our study identifies the C domain of the ECM protein TNC as a promising CAR T-cell therapy for pediatric solid tumors and brain tumors. While we focus here on pediatric cancer, our work has relevance to a broad range of adult cancers that express C.TNC.
Keywords: Chimeric antigen receptor - CAR; Immunotherapy; T cell.
© Author(s) (or their employer(s)) 2024. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: EW, SL, JW, TIS, GK, JZ, and SG have patent applications in the fields of cell or gene therapy for cancer. MB, GK, and SG are coinventors on a patent application for the developed Zip receptor technology. SG is a member of the Scientific Advisory Board of Be Biopharma and CARGO, and the Data and Safety Monitoring Board (DSMB) of Immatics and has received honoraria from TESSA Therapeutics within the last year. The other authors declare no competing interests.
Figures




References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous
