Biomaterials for in situ cell therapy
- PMID: 39574564
- PMCID: PMC11581612
- DOI: 10.1002/bmm2.12039
Biomaterials for in situ cell therapy
Abstract
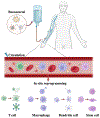
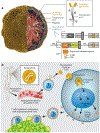
Cell therapy has revolutionized the treatment of various diseases, such as cancers, genetic disorders, and autoimmune diseases. Currently, most cell therapy products rely on ex vivo cell engineering, which requires sophisticated manufacturing processes and poses safety concerns. The implementation of in situ cell therapy holds the potential to overcome the current limitations of cell therapy and provides a broad range of applications and clinical feasibility in the future. A variety of biomaterials have been developed to improve the function and target delivery to specific cell types due to their excellent biocompatibility, tunable properties, and other functionalities, which provide a reliable method to achieve in vivo modulation of cell reprogramming. In this article, we summarize recent advances in biomaterials for in situ cell therapy including T cells, macrophages, dendritic cells, and stem cells reprogramming leveraging lipid nanoparticles, polymers, inorganic materials, and other biomaterials. Finally, we discuss the current challenges and future perspectives of biomaterials for in situ cell therapy.
Keywords: biomaterial; cell therapy; drug delivery; in situ.
Conflict of interest statement
CONFLICT OF INTEREST STATEMENT Yizhou Dong is a scientific advisory board member of Arbor Biotechnologies. The other authors declare no conflicts of interest.
Figures
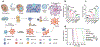


References
-
- Roth TL, Puig-Saus C, Yu R, Shifrut E, Carnevale J, Li PJ, Hiatt J, Saco J, Krystofinski P, Li H, Tobin V, Nguyen DN, Lee MR, Putnam AL, Ferris AL, Chen JW, Schickel J-N, Pellerin L, Carmody D, Alkorta-Aranburu G, del Gaudio D, Matsumoto H, Morell M, Mao Y, Cho M, Quadros RM, Gurumurthy CB, Smith B, Haugwitz M, Hughes SH, Weissman JS, Schumann K, Esensten JH, May AP, Ashworth A, Kupfer GM, Greeley SAW, Bacchetta R, Meffre E, Roncarolo MG, Romberg N, Herold KC, Ribas A, Leonetti MD, Marson A, Nature 2018, 559, 405. - PMC - PubMed
-
- Liu E, Marin D, Banerjee P, Macapinlac HA, Thompson P, Basar R, Nassif Kerbauy L, Overman B, Thall P, Kaplan M, Nandivada V, Kaur I, Nunez Cortes A, Cao K, Daher M, Hosing C, Cohen EN, Kebriaei P, Mehta R, Neelapu S, Nieto Y, Wang M, Wierda W, Keating M, Champlin R, Shpall EJ, Rezvani K, N. Engl. J. Med 2020, 382, 545. - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
