Switching of RNA splicing regulators in immature neuroblasts during adult neurogenesis
- PMID: 39576691
- PMCID: PMC11584179
- DOI: 10.7554/eLife.87083
Switching of RNA splicing regulators in immature neuroblasts during adult neurogenesis
Abstract
The lateral wall of the mouse subventricular zone harbors neural stem cells (NSC, B cells) which generate proliferating transient-amplifying progenitors (TAP, C cells) that ultimately give rise to neuroblasts (NB, A cells). Molecular profiling at the single-cell level struggles to distinguish these different cell types. Here, we combined transcriptome analyses of FACS-sorted cells and single-cell RNAseq to demonstrate the existence of an abundant, clonogenic and multipotent population of immature neuroblasts (iNB cells) at the transition between TAP and migrating NB (mNB). iNB are reversibly engaged in neuronal differentiation. Indeed, they keep molecular features of both undifferentiated progenitors, plasticity and unexpected regenerative properties. Strikingly, they undergo important progressive molecular switches, including changes in the expression of splicing regulators leading to their differentiation in mNB subdividing them into two subtypes, iNB1 and iNB2. Due to their plastic properties, iNB could represent a new target for regenerative therapy of brain damage.
Keywords: Subventricular zone; adult neurogenesis; immature neuroblasts; mouse; regenerative medicine; stem cells; transcriptomic analysis.
© 2023, Bernou et al.
Conflict of interest statement
CB, MM, MD, TK, BD, VB, SC, ND, VM, LB, CA, AC, FB No competing interests declared
Figures
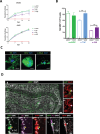

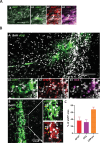





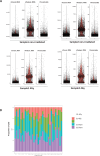









Update of
- doi: 10.1101/2023.03.12.532290
- doi: 10.7554/eLife.87083.1
- doi: 10.7554/eLife.87083.2
References
-
- Cebrian-Silla A, Nascimento MA, Redmond SA, Mansky B, Wu D, Obernier K, Romero Rodriguez R, Gonzalez-Granero S, García-Verdugo JM, Lim DA, Álvarez-Buylla A. Single-cell analysis of the ventricular-subventricular zone reveals signatures of dorsal and ventral adult neurogenesis. eLife. 2021;10:e67436. doi: 10.7554/eLife.67436. - DOI - PMC - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

