Protective effect of Dulaglutide, a GLP1 agonist, on acetic acid-induced ulcerative colitis in rats: involvement of GLP-1, TFF-3, and TGF-β/PI3K/NF-κB signaling pathway
- PMID: 39579211
- PMCID: PMC11985593
- DOI: 10.1007/s00210-024-03631-5
Protective effect of Dulaglutide, a GLP1 agonist, on acetic acid-induced ulcerative colitis in rats: involvement of GLP-1, TFF-3, and TGF-β/PI3K/NF-κB signaling pathway
Abstract
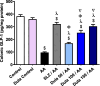
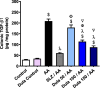
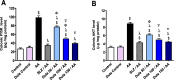
A chronic inflammatory condition of the colon called ulcerative colitis (UC) is characterized by mucosal surface irritation that extends from the rectum to the near proximal colon portions. The rationale of this work was to conclude if dulaglutide (Dula) could protect rats from developing colitis caused by exposure to acetic acid (AA). Rats were randomly divided into seven groups (each with eight rats): Normal control, Dula control, AA (received 2 milliliters of 3% v/v AA through the rectum), Sulfasalazine (SLZ); given SLZ (100 mg/kg) orally from day 11 to day 21 then AA intrarectally on day 22 and Dula groups ( pretreated with 50, 100 or 150 μg/kg subcutaneous injection of Dula - once weekly for three weeks and AA on day 22 to induce ulcerative colitis, colon tissues and blood samples were taken on day 23. By generating colonic histological deviations such as inflammatory processes, goblet cell death, glandular hyperplasia, and mucosa ulcers, Dula dropped AA-induced colitis. Additionally, these modifications diminished blood lactate dehydrogenase (LDH), C-reactive protein (CRP), colon weight, and the weight/length ratio of the colon. In addition, Dula decreased the oxidative stress biomarker malondialdehyde (MDA) and increased the antioxidant enzymes (total antioxidant capacity (TAC), reduced glutathione (GSH), and superoxide dismutase (SOD) concentrations). Dula also significantly reduced the expression of transforming growth factor-1 (TGF-β1), phosphatidylinositol-3-kinase (PI3K), protein kinase B (AKT) signaling pathway, and the inflammatory cytokines: nuclear factor kappa B (NF-κB), interleukin-6 (IL-6), and interferon-γ (IFN-γ) in colonic cellular structures. In addition, Dula enforced the levels of glucagon-like peptide-1 (GLP-1) and trefoil factor-3 (TFF-3) that were crucial to intestinal mucosa regeneration and healing of wounds. By modulating TGF-β1 in conjunction with other inflammatory pathways like PI3K/AKT and NF-κB, regulating the oxidant/antioxidant balance, and improving the integrity of the intestinal barrier, Dula prevented AA-induced colitis in rats.
Keywords: Dulaglutide; Phosphatidylinositol-3-kinase; Protein kinase B; Transforming growth factor-1; Trefoil factor-3; Ulcerative colitis.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests.
Figures










References
-
- Ala M, Jafari RM, Nematian H, Shadboorestan A, Dehpour AR (2022) Sodium selenite modulates IDO1/Kynurenine, TLR4, NF-κB and Bcl2/Bax pathway and mitigates acetic acid-induced colitis in rat. Cell Physiol Biochem 56:24–35 - PubMed
-
- Al-Dwairi A, Alqudah TE, Al-Shboul O, Alqudah M, Mustafa AG, Alfaqih MA (2019) Glucagon-like peptide-1 exerts anti-inflammatory effects on mouse colon smooth muscle cells through the cyclic adenosine monophosphate/nuclear factor-κB pathway in vitro [Corrigendum]. Journal of Inflammation Research 12:267–268 - PMC - PubMed
-
- Ali AA, Abd Al Haleem EN, Khaleel SA, Sallam AS (2017) Protective effect of cardamonin against acetic acid-induced ulcerative colitis in rats. Pharmacol Rep 69:268–275 - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

