The Secretion of Inflammatory Cytokines Triggered by TLR2 Through Calcium-Dependent and Calcium-Independent Pathways in Keratinocytes
- PMID: 39588538
- PMCID: PMC11588404
- DOI: 10.1155/mi/8892514
The Secretion of Inflammatory Cytokines Triggered by TLR2 Through Calcium-Dependent and Calcium-Independent Pathways in Keratinocytes
Abstract
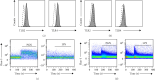
Keratinocytes can be activated by Cutibacterium acnes, leading to the production of proinflammatory cytokines via toll-like receptors (TLRs) 2 and 4. Although several studies have investigated keratinocytes, the mechanism of calcium-mediated activation remains unclear. Herein, we investigated whether calcium influx via TLR2 and TLR4 stimulation was involved in cytokine secretion by keratinocytes in HaCaT cells. Although TLR2 stimulation by peptidoglycan (PGN) increased intracellular calcium influx, TLR4 stimulation by lipopolysaccharide (LPS) did not increase it, as analyzed using flow cytometry with the calcium indicator Fluo-3. However, activation by either TLR2 or TLR4 ligands upregulated the intracellular calcium influx in THP-1 monocytes. Additionally, the expression of major proinflammatory cytokines and chemokines, such as interleukin (IL)-6, IL-8, IL-1α, granulocyte-macrophage colony-stimulating factor (GM-CSF), and monocyte chemoattractant protein-1 (MCP-1), was significantly increased by TLR2 in HaCaT cells. Moreover, treatment with the intracellular calcium chelator, BAPTA-AM, disrupted PGN-mediated induction of IL-6, IL-8, and MCP-1 production. Real-time quantitative polymerase chain reaction (PCR) and western blotting revealed that TLR2 stimulation induced expression of the epidermal differentiation marker keratin 1. In conclusion, TLR2-induced intracellular calcium influx plays a pivotal role in the secretion of proinflammatory cytokines, such as IL-6 and MCP-1, in keratinocytes. Moreover, the continuous influx of calcium via TLR2 activation leads to keratinization. In vitro studies using HaCaT cells provide basic research on the effect of TLR2-induced calcium on C. acnes-mediated inflammation in keratinocytes. These studies are limited in their ability to clinically predict what happens in human keratinocytes. Clinical studies on patients with acne, including three-dimensional (3D) cultures of primary keratinocytes, are required to develop new diagnostic markers for determining the severity of acne vulgaris.
Copyright © 2024 Eun-Ok Kim et al.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

