Axl and EGFR Dual-Specific Binding Affibody for Targeted Therapy in Nasopharyngeal Carcinoma
- PMID: 39594573
- PMCID: PMC11592995
- DOI: 10.3390/cells13221823
Axl and EGFR Dual-Specific Binding Affibody for Targeted Therapy in Nasopharyngeal Carcinoma
Abstract
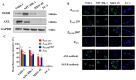
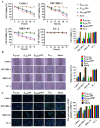
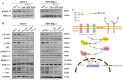
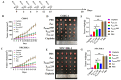
Nasopharyngeal carcinoma (NPC) is a tumor of the head and neck, with a higher incidence in southern China and Southeast Asia. Radiotherapy and chemotherapy are the main treatments; however, metastasis and recurrence remain the main causes of treatment failure. Further, the majority of patients are diagnosed in the late stage due to lack of tumor-specific biomarker for early diagnosis. Therefore, an effective treatment and early detection can improve the outcome of patient with NPC. Axl and EGFR are co-expressed in NPC tissues and play key roles in tumor proliferation, migration, and invasion, which are often correlated with poor prognosis and therapy resistance. In this study, we generated a novel bispecific affibody (Z239-1907) for the dual targeting and inhibition of Axl and EGFR expression in NPC-positive cells both in vitro and in vivo. The in vitro experiments demonstrated that Z239-1907 had more pronounced antitumor effects than either modality alone (ZAXL239 or ZEGFR1907) in NPC-positive cells. Further, mice bearing NPC-positive tumors showed significant inhibition in tumor growth after treatment with Z239-1907 compared to ZAXL239 and ZEGFR1907. The in vivo tumor targeting ability and imaging also showed that Z239-1907 specifically and selectively targeted NPC xenograft mice models and accumulate at tumor site as early as 30 min and disappeared within 24 h post-injection. Collectively, these results suggest that Z239-1907 dual-target affibody is a promising therapeutic agent and a molecular imaging probe for early diagnosis in NPC.
Keywords: Axl; EGFR; affibody molecules; nasopharyngeal carcinoma; targeted therapy.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



References
-
- Jacob J.B., Jacob M.K., Parajuli P. Review of immune checkpoint inhibitors in immuno-oncology. Adv. Pharmacol. 2021;91:111–139. - PubMed
-
- Burton E.M., Tawbi H.A. Bispecific Antibodies to PD-1 and CTLA4: Doubling Down on T Cells to Decouple Efficacy from Toxicity. Cancer Discov. 2021;11:1008–1010. doi: 10.1158/2159-8290.CD-21-0257. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

