Harnessing Bacterial Agents to Modulate the Tumor Microenvironment and Enhance Cancer Immunotherapy
- PMID: 39594765
- PMCID: PMC11593222
- DOI: 10.3390/cancers16223810
Harnessing Bacterial Agents to Modulate the Tumor Microenvironment and Enhance Cancer Immunotherapy
Abstract
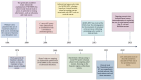
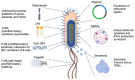
Cancer immunotherapy has revolutionized cancer treatment by leveraging the immune system to attack tumors. However, its effectiveness is often hindered by the immunosuppressive tumor microenvironment (TME), where a complex interplay of tumor, stromal, and immune cells undermines antitumor responses and allows tumors to evade immune detection. This review explores innovative strategies to modify the TME and enhance immunotherapy outcomes, focusing on the therapeutic potential of engineered bacteria. These bacteria exploit the unique characteristics of the TME, such as abnormal vasculature and immune suppression, to selectively accumulate in tumors. Genetically modified bacteria can deliver therapeutic agents, including immune checkpoint inhibitors and cytokines, directly to tumor sites. This review highlights how bacterial therapeutics can target critical immune cells within the TME, such as myeloid-derived suppressor cells and tumor-associated macrophages, thereby promoting antitumor immunity. The combination of bacterial therapies with immune checkpoint inhibitors or adoptive cell transfer presents a promising strategy to counteract immune suppression. Continued research in this area could position bacterial agents as a powerful new modality to reshape the TME and enhance the efficacy of cancer immunotherapy, particularly for tumors resistant to conventional treatments.
Keywords: antitumor immunity; bacterial therapeutics; cancer immunotherapy; engineered bacteria; immune checkpoint inhibitors; immunosuppression; tumor microenvironment.
Conflict of interest statement
P.d.F. and J.S. own equity in Tranquility Biodesign, LLC., a company committed to commercializing bacterial therapeutics.
Figures



References
-
- Ribas A. Adaptive Immune Resistance: How Cancer Protects from Immune Attack. Cancer Discov. 2015;5:915–919. doi: 10.1158/2159-8290.CD-15-0563. - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

