Supplementation of Oocytes by Microinjection with Extra Copies of mtDNA Alters Metabolite Profiles and Interactions with Expressed Genes in a Tissue-Specific Manner
- PMID: 39595653
- PMCID: PMC11591607
- DOI: 10.3390/biom14111477
Supplementation of Oocytes by Microinjection with Extra Copies of mtDNA Alters Metabolite Profiles and Interactions with Expressed Genes in a Tissue-Specific Manner
Abstract
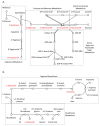
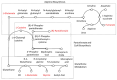
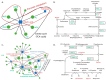
Mitochondrial DNA (mtDNA) supplementation can rescue poor oocyte quality and overcome embryonic arrest. Here, we investigated a series of sexually mature pigs generated through autologous and heterologous mtDNA supplementation. Brain, liver and heart tissues underwent metabolite profiling using gas chromatography-mass spectrometry and gene expression analysis through RNA-seq. They were then assessed for mRNA-metabolite interactions. The comparison between overall mtDNA supplemented and control pigs revealed that mtDNA supplementation reduced the lipids stearic acid and elaidic acid in heart tissue. However, heterologous mtDNA supplemented-derived pigs exhibited lower levels of abundance of metabolites when compared with autologous-derived pigs. In the brain, these included mannose, mannose 6-phosphate and fructose 6-phosphate. In the liver, maltose and cellobiose, and in the heart, glycine and glutamate were affected. mRNA-metabolite pathway analysis revealed a correlation between malate and CS, ACLY, IDH2 and PKLR in the liver and glutamate and PSAT1, PHGDH, CDO1 and ANPEP in the heart. Our outcomes demonstrate that mtDNA supplementation, especially heterologous supplementation, alters the metabolite and transcriptome profiles of brain, liver, and heart tissues. This is likely due to the extensive resetting of the balance between the nuclear and mitochondrial genomes in the preimplantation embryo, which induces a series of downstream effects.
Keywords: brain; gene expression; heart; liver; metabolic pathways; metabolite profile; mtDNA; mtDNA supplementation.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
-
- Cagnone G.L., Tsai T.S., Makanji Y., Matthews P., Gould J., Bonkowski M.S., Elgass K.D., Wong A.S., Wu L.E., McKenzie M., et al. Restoration of normal embryogenesis by mitochondrial supplementation in pig oocytes exhibiting mitochondrial DNA deficiency. Sci. Rep. 2016;6:23229. doi: 10.1038/srep23229. - DOI - PMC - PubMed
-
- Morimoto Y., Gamage U.S.K., Yamochi T., Saeki N., Morimoto N., Yamanaka M., Koike A., Miyamoto Y., Tanaka K., Fukuda A., et al. Mitochondrial Transfer into Human Oocytes Improved Embryo Quality and Clinical Outcomes in Recurrent Pregnancy Failure Cases. Int. J. Mol. Sci. 2023;24:2738. doi: 10.3390/ijms24032738. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

