Disruption of the Physical Interaction Between Carbonic Anhydrase IX and the Monocarboxylate Transporter 4 Impacts Lactate Transport in Breast Cancer Cells
- PMID: 39596062
- PMCID: PMC11593560
- DOI: 10.3390/ijms252211994
Disruption of the Physical Interaction Between Carbonic Anhydrase IX and the Monocarboxylate Transporter 4 Impacts Lactate Transport in Breast Cancer Cells
Abstract
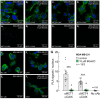
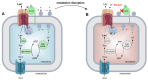
It has been previously established that breast cancer cells exhibit high expression of the monocarboxylate (lactate) transporters (MCT1 and/or MCT4) and carbonic anhydrase IX (CAIX) and form a functional metabolon for proton-coupled lactate export, thereby stabilizing intracellular pH. CD147 is the MCT accessory protein that facilitates the creation of the MCT/CAIX complex. This study describes how the small molecule Beta-Galactose 2C (BGal2C) blocks the physical and functional interaction between CAIX and either MCT1 or MCT4 in Xenopus oocytes, which reduces the rate of proton and lactate flux with an IC50 of ~90 nM. This value is similar to the Ki for inhibition of CAIX activity. Furthermore, it is shown that BGal2C blocks hypoxia-induced lactate transport in MDA-MB-231 and MCF-7 breast cancer cells, both of which express CAIX. As in oocytes, BGal2C interferes with the physical interaction between CAIX and MCTs in both cell types. Finally, X-ray crystallographic studies highlight unique interactions between BGal2C and a CAIX-mimic that are not observed within the CAII active site and which may underlie the strong specificity of BGal2C for CAIX. These studies demonstrate the utility of a novel sulfonamide in interfering with elevated proton and lactate flux, a hallmark of many solid tumors.
Keywords: BGal2C; MCT1/4; breast cancer; carbonic anhydrase IX; drug development; protein–drug interaction.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





References
-
- Sørlie T., Perou C.M., Tibshirani R., Aas T., Geisler S., Johnsen H., Hastie T., Eisen M.B., van de Rijn M., Jeffrey S.S., et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc. Natl. Acad. Sci. USA. 2001;98:10869–10874. doi: 10.1073/pnas.191367098. - DOI - PMC - PubMed
-
- Bauer K.R., Brown M., Cress R.D., Parise C.A., Caggiano V. Descriptive analysis of estrogen receptor (ER)-negative, progesterone receptor (PR)-negative, and HER2-negative invasive breast cancer, the so-called triple-negative phenotype: A population-based study from the California cancer Registry. Cancer. 2007;109:1721–1728. doi: 10.1002/cncr.22618. - DOI - PubMed
-
- Dent R., Trudeau M., Pritchard K.I., Hanna W.M., Kahn H.K., Sawka C.A., Lickley L.A., Rawlinson E., Sun P., Narod S.A. Triple-negative breast cancer: Clinical features and patterns of recurrence. Pt 1Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2007;13:4429–4434. doi: 10.1158/1078-0432.CCR-06-3045. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

