Molecular Roles of NADPH Oxidase-Mediated Oxidative Stress in Alzheimer's Disease: Isoform-Specific Contributions
- PMID: 39596364
- PMCID: PMC11594809
- DOI: 10.3390/ijms252212299
Molecular Roles of NADPH Oxidase-Mediated Oxidative Stress in Alzheimer's Disease: Isoform-Specific Contributions
Abstract
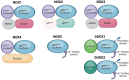
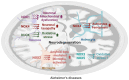
Oxidative stress is linked to the pathogenesis of Alzheimer's disease (AD), a neurodegenerative disorder marked by memory impairment and cognitive decline. AD is characterized by the accumulation of amyloid-beta (Aβ) plaques and the formation of neurofibrillary tangles (NFTs) of hyperphosphorylated tau. AD is associated with an imbalance in redox states and excessive reactive oxygen species (ROS). Recent studies report that NADPH oxidase (NOX) enzymes are significant contributors to ROS generation in neurodegenerative diseases, including AD. NOX-derived ROS aggravates oxidative stress and neuroinflammation during AD. In this review, we provide the potential role of all NOX isoforms in AD pathogenesis and their respective structural involvement in AD progression, highlighting NOX enzymes as a strategic therapeutic target. A comprehensive understanding of NOX isoforms and their inhibitors could provide valuable insights into AD pathology and aid in the development of targeted treatments for AD.
Keywords: Alzheimer’s disease; NADPH oxidases; oxidative stress; reactive oxygen species.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

