Co-Aggregation of TDP-43 with Other Pathogenic Proteins and Their Co-Pathologies in Neurodegenerative Diseases
- PMID: 39596445
- PMCID: PMC11594478
- DOI: 10.3390/ijms252212380
Co-Aggregation of TDP-43 with Other Pathogenic Proteins and Their Co-Pathologies in Neurodegenerative Diseases
Abstract
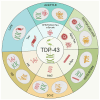
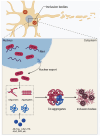
Pathological aggregation of a specific protein into insoluble aggregates is a common hallmark of various neurodegenerative diseases (NDDs). In the earlier literature, each NDD is characterized by the aggregation of one or two pathogenic proteins, which can serve as disease-specific biomarkers. The aggregation of these specific proteins is thought to be a major cause of or deleterious result in most NDDs. However, accumulating evidence shows that a pathogenic protein can interact and co-aggregate with other pathogenic proteins in different NDDs, thereby contributing to disease onset and progression synergistically. During the past years, more than one type of NDD has been found to co-exist in some individuals, which may increase the complexity and pathogenicity of these diseases. This article reviews and discusses the biochemical characteristics and molecular mechanisms underlying the co-aggregation and co-pathologies associated with TDP-43 pathology. The TDP-43 aggregates, as a hallmark of amyotrophic lateral sclerosis (ALS) and frontotemporal lobar degeneration (FTLD), can often be detected in other NDDs, such as Alzheimer's disease (AD), Parkinson's disease (PD), Huntington's disease (HD) and spinocerebellar ataxia type 2 (SCA2). In many cases, TDP-43 is shown to interact and co-aggregate with multiple pathogenic proteins in vitro and in vivo. Furthermore, the co-occurrence and co-aggregation of TDP-43 with other pathogenic proteins have important consequences that may aggravate the diseases. Thus, the current viewpoint that the co-aggregation of TDP-43 with other pathogenic proteins in NDDs and their relevance to disease progression may gain insights into the patho-mechanisms and therapeutic potential of various NDDs.
Keywords: TDP-43 pathology; co-aggregation; co-pathology; neurodegenerative disease; pathogenic protein.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

