Microbiome Shifts and Their Impact on Gut Physiology in Irritable Bowel Syndrome
- PMID: 39596460
- PMCID: PMC11594715
- DOI: 10.3390/ijms252212395
Microbiome Shifts and Their Impact on Gut Physiology in Irritable Bowel Syndrome
Abstract
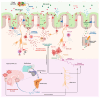
Irritable bowel syndrome (IBS) is one of the most prevalent functional gastrointestinal disorders characterized by recurrent abdominal pain and altered bowel habits. The exact pathophysiological mechanisms for IBS development are not completely understood. Several factors, including genetic predisposition, environmental and psychological influences, low-grade inflammation, alterations in gastrointestinal motility, and dietary habits, have been implicated in the pathophysiology of the disorder. Additionally, emerging evidence highlights the role of gut microbiota in the pathophysiology of IBS. This review aims to thoroughly investigate how alterations in the gut microbiota impact physiological functions such as the brain-gut axis, immune system activation, mucosal inflammation, gut permeability, and intestinal motility. Our research focuses on the dynamic "microbiome shifts", emphasizing the enrichment or depletion of specific bacterial taxa in IBS and their profound impact on disease progression and pathology. The data indicated that specific bacterial populations are implicated in IBS, including reductions in beneficial species such as Lactobacillus and Bifidobacterium, along with increases in potentially harmful bacteria like Firmicutes and Proteobacteria. Emphasis is placed on the imperative need for further research to delineate the role of specific microbiome alterations and their potential as therapeutic targets, providing new insights into personalized treatments for IBS.
Keywords: IBS; brain–gut axis; gut microbiota; irritable bowel syndrome; microbiome; pathophysiology; visceral hypersensitivity.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

