Neutrophil dynamics in pulmonary fibrosis: pathophysiological and therapeutic perspectives
- PMID: 39603661
- PMCID: PMC11600124
- DOI: 10.1183/16000617.0139-2024
Neutrophil dynamics in pulmonary fibrosis: pathophysiological and therapeutic perspectives
Abstract
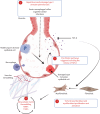
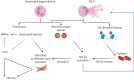
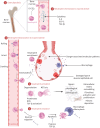
The shared pathobiological mechanisms driving progressive fibrosis in interstitial lung diseases (ILDs) remain unclear. Neutrophils, the most common immune cells in the human body, contain an extensive array of proteinases that are important for cell function, including tissue repair and remodelling. Increasing observational studies have reported elevated neutrophil counts in the respiratory tract and circulation of patients with ILD and suggest a role as a biomarker of disease severity. Neutrophils and their contents (including the formation of neutrophil extracellular traps (NETs)) are present in fibrotic lung tissue. Proteinases and NETs may drive fibrogenesis in animal and in vitro models and may impact transforming growth factor-β1 activation. However, the effect of neutrophil action, whether reparative or pathologically destructive to the delicate lung architecture, has yet to be determined. This review aims to summarise the current literature surrounding the potential role of the neutrophil as a biomarker and contributor to the pathogenesis of ILD. There is currently a paucity of treatment options in ILD driven by the knowledge gap underlying the overall disease mechanisms. This review concludes that neutrophils warrant further evaluation as manipulation of recruitment and function could provide a novel and much needed therapeutic strategy.
Copyright ©The authors 2024.
Conflict of interest statement
Conflict of interest: L.E. Crowley, R.A. Stockley, D.R. Thickett, A. Scott and D. Parekh have no perceived or potential conflicts of interest, financial or otherwise. D. Dosanjh is a full-time employee of AstraZeneca.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
