The Ercc1-/Δ mouse model of XFE progeroid syndrome undergoes accelerated retinal degeneration
- PMID: 39604117
- PMCID: PMC11896507
- DOI: 10.1111/acel.14419
The Ercc1-/Δ mouse model of XFE progeroid syndrome undergoes accelerated retinal degeneration
Abstract
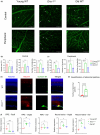
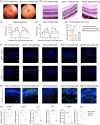
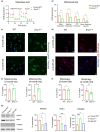
Age-related macular degeneration (AMD) is a major cause of vision loss in older adults. AMD is caused by degeneration in the macula of the retina. The retina is the highest oxygen consuming tissue in our body and is prone to oxidative damage. DNA damage is one hallmark of aging implicated in loss of organ function. Genome instability has been associated with several disorders that result in premature vision loss. We hypothesized that endogenous DNA damage plays a causal role in age-related retinal changes. To address this, we used a genetic model of systemic depletion of expression of the DNA repair enzyme ERCC1-XPF. The neural retina and retinal pigment epithelium (RPE) from Ercc1-/Δ mice, which models a human progeroid syndrome, were compared to age-matched wild-type (WT) and old WT mice. By 3-months-of age, Ercc1-/Δ mice presented abnormal optokinetic and electroretinogram responses consistent with photoreceptor dysfunction and visual impairment. Ercc1-/Δ mice shared many ocular characteristics with old WT mice including morphological changes, elevated DNA damage markers (γ-H2AX and 53BP1), and increased cellular senescence in the neural retinal and RPE, as well as pathological angiogenesis. The RPE is essential for the metabolic health of photoreceptors. The RPE from Ercc1-/Δ mice displayed mitochondrial dysfunction causing a compensatory glycolytic shift, a characteristic feature of aging RPE. Hence, our study suggests spontaneous endogenous DNA damage promotes the hallmarks of age-related retinal degeneration.
Keywords: DNA damage; age‐related retinal degeneration; cellular senescence.
© 2024 The Author(s). Aging Cell published by Anatomical Society and John Wiley & Sons Ltd.
Conflict of interest statement
None declared.
Figures



References
-
- Ahmad, A. , Robinson, A. R. , Duensing, A. , van Drunen, E. , Beverloo, H. B. , Weisberg, D. B. , Hasty, P. , Hoeijmakers, J. H. , & Niedernhofer, L. J. (2008). ERCC1‐XPF endonuclease facilitates DNA double‐strand break repair. Molecular and Cellular Biology, 28(16), 5082–5092. 10.1128/mcb.00293-08 - DOI - PMC - PubMed
-
- Bhagwat, N. , Olsen, A. L. , Wang, A. T. , Hanada, K. , Stuckert, P. , Kanaar, R. , D'Andrea, A. , Niedernhofer, L. J. , & McHugh, P. J. (2009). XPF‐ERCC1 participates in the Fanconi anemia pathway of cross‐link repair. Molecular and Cellular Biology, 29(24), 6427–6437. 10.1128/mcb.00086-09 - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

