Biomarkers of response to ocrelizumab in relapsing-remitting multiple sclerosis
- PMID: 39606235
- PMCID: PMC11600310
- DOI: 10.3389/fimmu.2024.1480676
Biomarkers of response to ocrelizumab in relapsing-remitting multiple sclerosis
Erratum in
-
Corrigendum: Biomarkers of response to ocrelizumab in relapsing-remitting multiple sclerosis.Front Immunol. 2024 Dec 19;15:1535051. doi: 10.3389/fimmu.2024.1535051. eCollection 2024. Front Immunol. 2024. PMID: 39749346 Free PMC article.
Abstract
Objective: To ascertain the changes of serum neurofilament light chain (sNfL) and glial fibrillary acidic protein (sGFAP) values in relapsing-remitting multiple sclerosis (RRMS) patients treated with ocrelizumab and their association with treatment response.
Methods: Multicenter prospective study including 115 RRMS patients initiating ocrelizumab treatment between February 2020 and March 2022 followed during a year. Serum samples were collected at baseline and every 3 months to measure sNfL and sGFAP levels using single-molecule array (SIMOA) technology. Based on age and body mass index, sNfL values were standardized using z-score. NEDA (non-evidence of disease activity)-3 status was defined for patients free of disease activity after a year of follow-up. Inflammation (INFL) was considered when new relapses occurred during follow-up or new MRI lesions were found at 1-year exploration. PIRA (progression independent of relapse activity) was defined as disability progression occurring in the absence of relapses or new MRI activity.
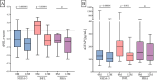
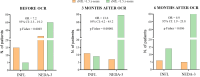
Results: After a year on ocrelizumab, 85 patients (73.9%) achieved NEDA-3. Thirty patients did not achieve NEDA: 20 (17.4%) because of INFL and 10 (8.7%) because of PIRA. Of INFL patients, 6 (30.0%) had relapses, and 17 (85.0%) had at least one new MRI lesion at the 12-month examination. At baseline, INFL patients had higher sNfL (p = 0.0003) and sGFAP (p = 0.03) than the NEDA-3 group. PIRA patients mostly exhibited low sNfL and heterogeneous sGFAP levels. After a year, NEDA-3 and INFL patients showed similar decreases in sNfL (p < 0.0001) and sGFAP (p < 0.0001 for NEDA-3 and p = 0.001 for INFL ones). However, the decrease occurred earlier in NEDA-3 patients. Accordingly, sNfL > 1.5 z-score 3 months after ocrelizumab initiation indicated a higher risk of inflammation (OR = 13.6; p < 0.0001). Decrease in sGFAP values occurred later in both groups, with significant reductions observed at 12 months for INFL and 6 and 12 months for NEDA-3. No significant changes in sNfL or sGFAP were observed in PIRA patients.
Conclusion: Ocrelizumab induced normalization of sNfL and sGFAP in the majority of NEDA-3 and inflammatory patients but did not cause changes in the PIRA group. Our data suggest that normalization of sNfL and sGFAP is associated with the lack of inflammatory-associated disease progression but it may not affect non-inflammatory PIRA.
Keywords: glial fibrillary acidic protein; multiple sclerosis; neurofilament light chain; ocrelizumab; serum biomarkers.
Copyright © 2024 Rodríguez-Jorge, Fernández-Velasco, Villarrubia, Gracia-Gil, Fernández, Meca-Lallana, Díaz-Pérez, Sainz de la Maza, Pacheco, Quiroga, Ramió-Torrentà, Martínez-Yélamos, Bau, Monreal, López-Real, Rodero-Romero, Borrega, Díaz, Eguía, Espiño, Chico-García, Barrero, Martínez-Ginés, García-Domínguez, De la Fuente, Moreno, Sainz-Amo, Mañé-Martínez, Caminero, Castellanos, Gómez López, Labiano-Fontcuberta, Ayuso, Abreu, Hernández, Meca-Lallana, Martín-Aguilar, Muriel García, Masjuan, Costa-Frossard and Villar.
Conflict of interest statement
FR-J received research grants and travel support for speaking engagements from Janssen, Biogen, Novartis, Roche, Sanofi-Genzyme, Bristol-Myers-Squibb and Merck. JG-G received research support, compensation for participating on advisory boards, lecture fees, and/or travel support from Almirall, Bayer, Biogen, Genzyme‐Sanofi, Novartis, Roche, and Teva. EF received research support, compensation for participating on advisory boards, speaking fees, and/or funding for travel from Almirall, Bayer, Biogen, Genzyme‐Sanofi, Merck, Novartis, and Roche. VM-L received consulting and speaking fees from Almirall, Biogen, Genzyme, Janssen, Merck, Novartis, Roche, Terumo, Sanofi, Teva, and Bristol Myers Squibb. CD-P received funding for training and scientific meetings from Sanofi, Merck, Novartis and Roche. SS received research grants, travel support or honoraria for speaking engagements from Almirall, Biogen, Bristol Myers Squibb, Janssen, Merck, Novartis, Roche, Sanofi-Genzyme, and Teva. AQ is funded by a grant from the Fundación Francisco Soria y Melguizo and has received funding from Merck, Novartis, and Horizon Therapeutics to attend conferences. LR-T received compensation for consulting services and speaking fees from Biogen, Novartis, Bayer, Merck, Sanofi, Genzyme, Roche, Bristol-Myers-Squibb, TEVA, and Horizon. SM-Y received honoraria for participating on advisory boards and for collaborations as consultant and scientific communications and also received research support as well as funding for travel and congress expenses from Roche, Biogen Idec, Novartis, TEVA, Merck, Genzyme, Sanofi, Bayer, Almirall, and Bristol Myers Squibb. EM received research grants, travel support, or honoraria for speaking engagements from Almirall, Merck, Roche, Sanofi, Bristol Myers Squibbb, Biogen, Janssen, and Novartis. AL-R received speaker and consultation fees from Biogen, Janssen, Novartis, Roche and Sanofi, and congress travel support from Roche. LBo received research grants and travel support from Merck, Roche, Novartis, Sanofi, Horizon and Bristol Myer Squibb. JC-G received speaker fees, travel support, and/or served on advisory boards by Biogen, Sanofi, Bayer, Janssen, BMS, and Bial. FB received compensation for consulting services and speaking honoraria from Almirall, Biogen, Bristol Myer Squibb, Genzyme, Johnson & Johnson, Merck, Novartis, Roche, Sanofi, Teva. MM-G received compensation for consulting services and speaking fees from Merck, Biogen, Novartis, Sanofi-Genzyme, Almirall, BMS, Janssen, Roche, Horizon, and Viatris. JG-D received honoraria as speaker, advisor and researcher from Almirall, Bristol Myers Squibb, Biogen, Janssen, Merck, Novartis, Roche, Teva, and Sanofi. MM-M received research support, funding for travel and congress expenses from Biogen Idec, Teva Pharmaceutical Industries Ltd., Sanofi-Aventis, Merck Serono, Novartis, Bayer Schering Pharma, Bristol Myers Squibb and Roche. JM-L received honoraria as a consultant, lecturer in meetings and has participated in clinical trials and other research projects promoted by Alexion, Almirall, Biogen, Bristol-Meyers-Squibb, Horizon, Johnson & Johnson, Merck, Neuraxpharm, Novartis, Roche, Sandoz, Sanofi and UCB. LC-F received speaker fees, travel support, and/or served on advisory boards by Biogen, Sanofi, Merck, Bayer, Novartis, Roche, Teva, Celgene, Ipsen, Biopas, Almirall. LV received research grants and personal fees from Merck, Roche, Sanofi, Bristol Myers Squibb, Biogen, and Novartis. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Figures


References
-
- Benkert P, Meier S, Schaedelin S, Manouchehrinia A, Yaldizli Ö, Maceski A, et al. . Serum neurofilament light chain for individual prognostication of disease activity in people with multiple sclerosis: a retrospective modelling and validation study. Lancet Neurol. (2022) 21:246–57. doi: 10.1016/S1474-4422(22)00009-6 - DOI - PubMed
-
- Monreal E, Fernández-Velasco JI, García-Sánchez MI, Sainz de la Maza S, Llufriu S, Álvarez-Lafuente R, et al. . Association of serum neurofilament light chain levels at disease onset with disability worsening in patients with a first demyelinating multiple sclerosis event not treated with high-efficacy drugs. JAMA Neurol. (2023) 80:397–403. doi: 10.1001/jamaneurol.2023.0010 - DOI - PMC - PubMed
-
- Barro C, Healy BC, Liu Y, Saxena S, Paul A, Polgar-Turcsanyi M, et al. . Serum GFAP and NfL levels differentiate subsequent progression and disease activity in patients with progressive multiple sclerosis. Neurol Neuroimmunol Neuroinflamm. (2022) 10:e200052. doi: 10.1212/NXI.0000000000200052 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

