Raphe and ventrolateral medulla proteomics in sudden unexplained death in childhood with febrile seizure history
- PMID: 39607506
- PMCID: PMC11604820
- DOI: 10.1007/s00401-024-02832-9
Raphe and ventrolateral medulla proteomics in sudden unexplained death in childhood with febrile seizure history
Abstract
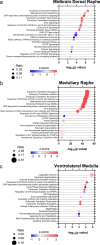
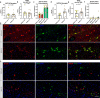
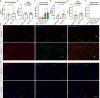
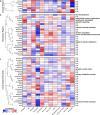
Sudden unexplained death in childhood (SUDC) is death of a child ≥ 12 months old that is unexplained after autopsy and detailed analyses. Among SUDC cases, ~ 30% have febrile seizure (FS) history, versus 2-5% in the general population. SUDC cases share features with sudden unexpected death in epilepsy (SUDEP) and sudden infant death syndrome (SIDS), in which brainstem autonomic dysfunction is implicated. To understand whether brainstem protein changes are associated with FS history in SUDC, we performed label-free quantitative mass spectrometry on microdissected midbrain dorsal raphe, medullary raphe, and the ventrolateral medulla (n = 8 SUDC-noFS, n = 11 SUDC-FS). Differential expression analysis between SUDC-FS and SUDC-noFS at p < 0.05 identified 178 altered proteins in dorsal raphe, 344 in medullary raphe, and 100 in the ventrolateral medulla. These proteins were most significantly associated with increased eukaryotic translation initiation (p = 3.09 × 10-7, z = 1.00), eukaryotic translation elongation (p = 6.31 × 10-49, z = 6.01), and coagulation system (p = 1.32 × 10-5, z = 1.00). The medullary raphe had the strongest enrichment for altered signaling pathways, including with comparisons to three other brain regions previously analyzed (frontal cortex, hippocampal dentate gyrus, cornu ammonus). Immunofluorescent tissue analysis of serotonin receptors identified 2.1-fold increased 5HT2A in the medullary raphe of SUDC-FS (p = 0.025). Weighted gene correlation network analysis (WGCNA) of case history indicated that longer FS history duration significantly correlated with protein levels in the medullary raphe and ventrolateral medulla; the most significant gene ontology biological processes were decreased cellular respiration (p = 9.8 × 10-5, corr = - 0.80) in medullary raphe and decreased synaptic vesicle cycle (p = 1.60 × 10-7, corr = - 0.90) in the ventrolateral medulla. Overall, FS in SUDC was associated with more protein differences in the medullary raphe and was related with increased translation-related signaling pathways. Future studies should assess whether these changes result from FS or may in some way predispose to FS or SUDC.
Keywords: Febrile seizures; Laser capture microdissection; Proteomics; SUDC.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Conflict of interest: The authors declare no competing interests.
Figures




References
-
- Ackerman MJ, Andrew TA, Baker AM, Devinsky O, Downs JC, Keens T et al (2016) An association of hippocampal malformations and sudden death? We need more data. Forensic Sci Med Pathol 12:229–231. 10.1007/s12024-016-9765-1 - PubMed
-
- Aira Z, Buesa I, Gallego M, García del Caño G, Mendiable N, Mingo J et al (2012) Time-dependent cross talk between spinal serotonin 5-HT2A receptor and mGluR1 subserves spinal hyperexcitability and neuropathic pain after nerve injury. J Neurosci 32:13568–13581. 10.1523/JNEUROSCI.1364-12.2012 - PMC - PubMed
-
- Bagdy G, Kecskemeti V, Riba P, Jakus R (2007) Serotonin and epilepsy. J Neurochem 100:857–873. 10.1111/j.1471-4159.2006.04277.x - PubMed
-
- Benarroch EE (2014) Medullary serotonergic system: organization, effects, and clinical correlations. Neurology 83:1104–1111. 10.1212/WNL.0000000000000806 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

