Management of autosomal dominant hypocalcemia type 1: Literature review and clinical practice recommendations
- PMID: 39607645
- PMCID: PMC11950097
- DOI: 10.1007/s40618-024-02496-y
Management of autosomal dominant hypocalcemia type 1: Literature review and clinical practice recommendations
Abstract
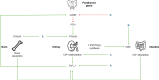
Purpose: Autosomal Dominant Hypocalcemia type 1 (ADH1), caused by gain-of-function variants in the calcium-sensing receptor (CASR), is characterized by a variable degree of hypocalcemia and hypercalciuria with inappropriately low PTH. The clinical spectrum is broad, ranging from being asymptomatic to presenting with severe clinical features of hypocalcemia and end-organ damage such as nephrolithiasis and intracerebral calcifications. Although the underlying pathophysiology is different, ADH1 patients are often managed as patients with 'classical' primary hypoparathyroidism, possibly leading to (exacerbation of) hypercalciuria. New treatments such as PTH analogues and calcilytics directly targeting the CASR are in the pipeline. Specific clinical guidance for treatment and monitoring of ADH1 patients is lacking. The purpose of this study is to provide a literature review on management of ADH1, including new therapies, and to formulate practice recommendations.
Methods: We searched for articles and ongoing clinical trials regarding management of ADH1.
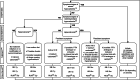
Results: Forty articles were included. First we review the conventional treatment of ADH1, focusing on active vitamin D, calcium supplements, thiazide diuretics, phosphorus binders and dietary recommendations. In a second part we give an overview of studies with emerging treatments in ADH1: PTH analogues (PTH1-34, rhPTH1-84, TransCon PTH and others) and calcilytics (preclinical studies and clinical trials). In a third part we discuss literature findings regarding monitoring of ADH1 patients. Finally, we formulate clinical practice recommendations.
Conclusion: We provide an overview of conventional and new treatments for ADH1 patients. Based on these data, we propose practical recommendations to assist clinicians in the management of ADH1 patients.
Keywords: Autosomal Dominant Hypocalcemia type 1; Calcilytics; Calcium sensing receptor; Parathyroid hormone analogues; Practice recommendations.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Conflict of interest: We report no conflict of interest.
Figures

References
-
- A new mutation in the calcium-sensing receptor gene causing hypocalcaemia: case report of a father and two sons - PubMed. https://pubmed.ncbi.nlm.nih.gov/28741586/. Accessed 10 Apr 2022 - PubMed
-
- Kinoshita Y, Hori M, Taguchi M et al (2014) Functional activities of mutant calcium-sensing receptors determine clinical presentations in patients with autosomal dominant hypocalcemia. J Clin Endocrinol Metab 99. 10.1210/JC.2013-3430 - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources

