Characterizing the pharmacological interaction of the antimalarial combination artefenomel-piperaquine in healthy volunteers with induced blood-stage Plasmodium falciparum to predict efficacy in patients with malaria
- PMID: 39609822
- PMCID: PMC11603672
- DOI: 10.1186/s12916-024-03787-0
Characterizing the pharmacological interaction of the antimalarial combination artefenomel-piperaquine in healthy volunteers with induced blood-stage Plasmodium falciparum to predict efficacy in patients with malaria
Abstract
Background: The combination antimalarial artefenomel-piperaquine failed to achieve target efficacy in a phase 2b study in Africa and Vietnam. We retrospectively evaluated whether characterizing the pharmacological interaction of this antimalarial combination in a volunteer infection study (VIS) would have enabled prediction of the phase 2b study results.
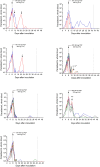
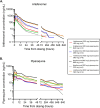
Methods: Twenty-four healthy adults enrolled over three consecutive cohorts were inoculated with Plasmodium falciparum-infected erythrocytes on day 0. Participants were randomized within each cohort to one of seven dose combination groups and administered a single oral dose of artefenomel-piperaquine on day 8. Participants received definitive antimalarial treatment with artemether-lumefantrine upon parasite regrowth or on day 42 ± 2. The general pharmacodynamic interaction (GPDI) model implemented in the Bliss Independence additivity criterion was developed to characterize the pharmacological interaction between artefenomel and piperaquine. Simulations based on the model were performed to predict the outcomes of the phase 2b combination study.
Results: For a dose of 800 mg artefenomel administered with 640 mg, 960 mg, or 1440 mg piperaquine, the simulated adequate parasitological response at day 28 (APR28), incorporating actual patient pharmacokinetic (PK) data from the phase 2b trial, was 69.4%, 63.9%, and 74.8%, respectively. These results closely matched the observed APR28 in the phase 2b trial of 67.0%, 65.5%, and 75.4%, respectively.
Conclusions: These results indicate that VIS offer an efficient means for informing antimalarial combination trials conducted in the field, potentially expediting clinical development.
Trial registration: This study was registered on ClinicalTrials.gov on 11 May 2018 with registration number NCT03542149.
Keywords: Antimalarial; Artefenomel; Combination; Pharmacodynamics; Pharmacokinetics; Piperaquine; Volunteer infection study.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: The study was approved by the QIMR Berghofer Medical Research Institute Human Research Ethics Committee (reference number: P2370). All participants gave written informed consent before enrolment. Consent for publication: Not applicable. Competing interests: JJM, MEG, and SC are currently employed by Medicines for Malaria Venture (MMV) which funded the study. BEB and JSM received funding from MMV to perform the study. All other authors declare no competing interests.
Figures

References
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

