Socio-affective communication in Tph2-deficient rat pups: communal nesting aggravates growth retardation despite ameliorating maternal affiliation deficits
- PMID: 39614401
- PMCID: PMC11606121
- DOI: 10.1186/s13229-024-00629-x
Socio-affective communication in Tph2-deficient rat pups: communal nesting aggravates growth retardation despite ameliorating maternal affiliation deficits
Abstract
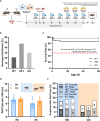
Background: A lack of serotonin (also known as 5-hydroxytryptamine, 5-HT) in the brain due to deficiency of the rate-limiting enzyme in 5-HT synthesis, tryptophan hydroxylase 2 (TPH2), was recently reported to result in impaired maternal affiliation across species, including mice, rats, and monkeys. In rodents, this was reflected in a lack of preference for maternal odors and reduced levels of isolation-induced ultrasonic vocalizations (USV), possibly contributing to a severe growth retardation phenotype.
Methods: Here, we tested whether growth retardation, maternal affiliation deficits, and/or impairments in socio-affective communication caused by Tph2 deficiency can be rescued through early social enrichment in rats. To this aim, we compared male and female Tph2-/- knockout and Tph2+/- heterozygous rat pups to Tph2+/+ wildtype littermate controls, with litters being randomly assigned to standard nesting (SN; one mother with her litter) or communal nesting (CN; two mothers with their two litters).
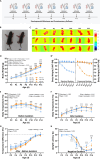
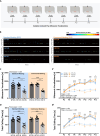
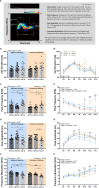
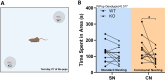
Results: Our results show that Tph2 deficiency causes severe growth retardation, together with moderate impairments in somatosensory reflexes and thermoregulatory capabilities, partially aggravated by CN. Tph2 deficiency further led to deficits in socio-affective communication, as evidenced by reduced emission of isolation-induced USV, associated with changes in acoustic features, clustering of subtypes, and temporal organization. Although CN did not rescue the impairments in socio-affective communication, CN ameliorated the maternal affiliation deficit caused by Tph2 deficiency in the homing test. To close the communicative loop between mother and pup, we assessed maternal preference and showed that mothers display a preference for Tph2+/+ controls over Tph2-/- pups, particularly under CN conditions. This is consistent with the aggravated growth phenotype in Tph2-/- pups exposed to the more competitive CN environment.
Conclusion: Together, this indicates that CN aggravates growth retardation despite ameliorating maternal affiliation deficits in Tph2-deficient rat pups, possibly due to reduced and acoustically altered isolation-induced USV, hindering efficient socio-affective communication between mother and pup.
Keywords: Autism; Neurodevelopmental disorders; Serotonin; Social behavior; Tryptophan hydroxylase 2; Ultrasonic vocalizations.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests.
Figures

Similar articles
-
Tryptophan hydroxylase 2 deficiency alters autism-related behavioural phenotypes in rats.Sci Rep. 2025 Jul 1;15(1):20522. doi: 10.1038/s41598-025-05684-9. Sci Rep. 2025. PMID: 40594285 Free PMC article.
-
Tph2 deficiency leads to alterations in social adjustment and socio-affective communication in neonatal rats: No rescue effect of communal nesting.Prog Neuropsychopharmacol Biol Psychiatry. 2025 Aug 12;141:111469. doi: 10.1016/j.pnpbp.2025.111469. Online ahead of print. Prog Neuropsychopharmacol Biol Psychiatry. 2025. PMID: 40812712
-
Reduced isolation-induced pup ultrasonic communication in mouse pups lacking brain serotonin.Mol Autism. 2015 Mar 8;6:13. doi: 10.1186/s13229-015-0003-6. eCollection 2015. Mol Autism. 2015. PMID: 25901271 Free PMC article.
-
Affective communication in rodents: serotonin and its modulating role in ultrasonic vocalizations.Behav Pharmacol. 2015 Sep;26(6):506-21. doi: 10.1097/FBP.0000000000000172. Behav Pharmacol. 2015. PMID: 26221830 Review.
-
Pro-social ultrasonic communication in rats: insights from playback studies.J Neurosci Methods. 2014 Aug 30;234:73-81. doi: 10.1016/j.jneumeth.2014.01.023. Epub 2014 Feb 4. J Neurosci Methods. 2014. PMID: 24508146 Review.
Cited by
-
Central Serotonin Deficiency Impairs Recovery of Sensorimotor Abilities After Spinal Cord Injury in Rats.Int J Mol Sci. 2025 Mar 19;26(6):2761. doi: 10.3390/ijms26062761. Int J Mol Sci. 2025. PMID: 40141402 Free PMC article.
-
Tryptophan hydroxylase 2 deficiency alters autism-related behavioural phenotypes in rats.Sci Rep. 2025 Jul 1;15(1):20522. doi: 10.1038/s41598-025-05684-9. Sci Rep. 2025. PMID: 40594285 Free PMC article.
References
-
- Aerts T, Boonen A, Geenen L, Stulens A, Masin L, Pancho A, Francis A, Pepermans E, Baggerman G, Van Roy F, Wöhr M, Seuntjens E. Altered socio-affective communication and amygdala development in mice with protocadherin10-deficient interneurons. Open Biol. 2024;14(6):240113. 10.1098/rsob.240113. - PMC - PubMed
-
- Allin JT, Banks EM. Functional aspects of ultrasound production by infant albino rats (Rattus norvegicus). Anim Behav. 1972;20(1):175–85. 10.1016/s0003-3472(72)80189-1. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

