Restoration of hair follicle inductive properties by depletion of senescent cells
- PMID: 39614601
- PMCID: PMC11709086
- DOI: 10.1111/acel.14353
Restoration of hair follicle inductive properties by depletion of senescent cells
Abstract
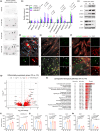
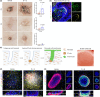
Senescent cells secrete a senescence-associated secretory phenotype (SASP), which can induce senescence in neighboring cells. Human dermal papilla (DP) cells lose their original hair inductive properties when expanded in vitro, and rapidly accumulate senescent cells in culture. Protein and RNA-seq analysis revealed an accumulation of DP-specific SASP factors including IL-6, IL-8, MCP-1, and TIMP-2. We found that combined senolytic treatment of dasatinib and quercetin depleted senescent cells, and reversed SASP accumulation and SASP-mediated repressive interactions in human DP culture, resulting in an increased Wnt-active cell population. In hair reconstitution assays, senolytic-depleted DP cells exhibited restored hair inductive properties by regenerating de novo hair follicles (HFs) compared to untreated DP cells. In 3D skin constructs, senolytic-depleted DP cells enhanced inductive potential and hair lineage specific differentiation of keratinocytes. These data revealed that senolytic treatment of cultured human DP cells markedly increased their inductive potency in HF regeneration, providing a new rationale for clinical applications of senolytic treatment in combination with cell-based therapies.
Keywords: cellular senescence; dasatinib; dermal papilla; hair follicle; quercetin; regeneration; senescence‐associated secretory phenotype; senolytic.
© 2024 The Author(s). Aging Cell published by Anatomical Society and John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflict of interest. Columbia University has filed patents on the use of human DP cells in HF neogenesis.
Figures


References
-
- Abaci, H. E. , Coffman, A. , Doucet, Y. , Chen, J. , Jackow, J. , Wang, E. , Guo, Z. , Shin, J. U. , Jahoda, C. A. , & Christiano, A. M. (2018). Tissue engineering of human hair follicles using a biomimetic developmental approach. Nature Communications, 9(1), 5301. 10.1038/s41467-018-07579-y - DOI - PMC - PubMed
-
- Acosta, J. C. , Banito, A. , Wuestefeld, T. , Georgilis, A. , Janich, P. , Morton, J. P. , Athineos, D. , Kang, T. W. , Lasitschka, F. , Andrulis, M. , Pascual, G. , Morris, K. J. , Khan, S. , Jin, H. , Dharmalingam, G. , Snijders, A. P. , Carroll, T. , Capper, D. , Pritchard, C. , … Gil, J. (2013). A complex secretory program orchestrated by the inflammasome controls paracrine senescence. Nature Cell Biology, 15(8), 978–990. 10.1038/ncb2784 - DOI - PMC - PubMed
-
- Baar, M. P. , Brandt, R. M. C. , Putavet, D. A. , Klein, J. D. D. , Derks, K. W. J. , Bourgeois, B. R. M. , Stryeck, S. , Rijksen, Y. , van Willigenburg, H. , Feijtel, D. A. , van der Pluijm, I. , Essers, J. , van Cappellen, W. A. , van , I. W. F. , Houtsmuller, A. B. , Pothof, J. , de Bruin, R. W. F. , Madl, T. , Hoeijmakers, J. H. J. , … de Keizer, P. L. J. (2017). Targeted apoptosis of senescent cells restores tissue homeostasis in response to chemotoxicity and aging. Cell, 169(1), 132–147.e116. 10.1016/j.cell.2017.02.031 - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

