Hepatic real-world outcomes with obeticholic acid in primary biliary cholangitis (HEROES): A trial emulation study design
- PMID: 39630028
- PMCID: PMC12077331
- DOI: 10.1097/HEP.0000000000001174
Hepatic real-world outcomes with obeticholic acid in primary biliary cholangitis (HEROES): A trial emulation study design
Abstract
Background and aims: Primary biliary cholangitis is a rare, progressive liver disease. Obeticholic acid (OCA) received accelerated approval for treating patients with primary biliary cholangitis in whom ursodeoxycholic acid failed, based on a surrogate endpoint of reduction in ALP. Analysis of the long-term safety extension with 2 external control groups demonstrated a significant increase in event-free survival in OCA-treated patients. This fully real-world evidence study assessed the effect of OCA treatment on clinical outcomes.
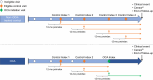
Approach and results: This trial emulation used data from the Komodo Healthcare Map claims database linked to US national laboratory, transplant, and death databases. Patients with compensated primary biliary cholangitis and intolerance/inadequate response to ursodeoxycholic acid who initiated OCA therapy were compared with patients who were OCA-eligible but not OCA-treated. The primary endpoint was time to the first occurrence of death, liver transplant, or hospitalization for hepatic decompensation, analyzed using a propensity-score weighted Cox proportional hazards model. Baseline prognostic factors were balanced using standardized morbidity ratio weighting. For the primary analysis, 4174 patients contributed 11,246 control index dates, and 403 patients contributed OCA indexes. Weighted groups were well balanced. Median (95% CI) follow-up in the OCA and non-OCA arms was 9.3 (8.4-10.6) months and 17.5 (16.2-18.6) months (weighted population; censored at discontinuation). Eight events occurred in the OCA arm and 32 in the weighted control (HR = 0.37; 95% CI = 0.14-0.75; p < 0.001). Effects were consistent for each component of the composite endpoint.
Conclusions: We identified a 63% reduced risk of hospitalization for hepatic decompensation, liver transplant, or death in OCA-treated versus non-OCA-treated individuals.
Trial registration: HEROES; ClinicalTrials.gov NCT05292872.
Keywords: confirmatory trial; death; hepatic decompensation; liver disease; liver transplant.
Copyright © 2024 The Author(s). Published by Wolters Kluwer Health, Inc.
Conflict of interest statement
M. Alan Brookhart advises and owns stock in Target RWE. He advises Amgen, Astellas/Seagen, Atara Biotherapeutics, Brigham and Women’s Hospital, Kite, Gilead, NIDDK, Vertex, AccompanyHealth, Target RWE, and Vertex. He advises and was employed by Intercept at the time they study was conducted. He owns stock in Accompany Health. Tracy J. Mayne was employed by Intercept at the time the study was conducted. Charles Coombs owns stock in and is employed by Syneos Health and Walmart. Alexander Breskin owns stock in and is employed by Regeneron Pharmaceuticals and Target RWE. Erik Ness was employed by Intercept at the time the study was conducted. He is employed by Madrigal. Leona Bessonova is employed by Intercept. Yucheng Julia Chu consults for and is employed by Intercept. Jing Li is employed by Intercept. Michael W. Fried is employed by and owns stock in Target RWE. Bettina E. Hansen consults for, is on the speakers’ bureau, and received grants from Mirum. She consults for, advises, and received grants from Cymabay, Intercept, and Ipsen. She consults for and advises Pliant. She consults for and received grants from Albireo, Calliditas, and Eiger. She consults for Enyo. She received grants from Gilead. Kris V. Kowdley has received honoraria, fees, equity, research support, and clinical trial grants from AbbVie, Corcept, CymaBay, Enanta, Genfit, Gilead, GSK, Hanmi, HighTide, Inipharm, Intercept, Madrigal, Mirum, Novo Nordisk, NGM Bio, Pfizer, Pliant, Terns, Viking, and 89bio. He advises and received grants from Boehringer Ingelheim and Ipsen. He advises Arbormed. He received grants from AstraZeneca and Akero. He receives royalties from UpToDate. David Jones consults for, is on the speakers’ bureau for, and received grants from Advanz and Intercept. He consults for and is on the speakers’ bureau for Abbott, Ipsen, and GSK. He consults for and received grants from Intercept. He consults for Cymabay and Umecrine. He is on the speakers’ bureau for Falk. George Mells received grants from Intercept. Palak J. Trivedi receives institutional salary support from the National Institute for Health Research (NIHR) Birmingham Biomedical Research Centre (BRC). [This paper presents independent research supported by the Birmingham NIHR BRC based at the University Hospitals Birmingham National Health Service Foundation Trust and the University of Birmingham. The views expressed are those of the author(s) and not necessarily those of the National Health Service, the NIHR, or the Department of Health.] Palak J. Trivedi has received grant support from Wellcome Trust, Medical Research Foundation, the LifeArc Foundation, Innovate UK, GSK, Guts UK, PSC Support, Intercept, Falk, Gilead, and Bristol Myers Squibb; has received speaker fees from Intercept and Falk; and has been an advisory board member/consultant for Albireo, CymaBay, Intercept, Ipsen, Falk, GSK, and Pliant. He consults for, advises, is on the speakers’ bureau, and received grants from Advanz. Shaun Hiu received grants from Intercept. James Wason received grants from Intercept. Rachel Smith received grants from Advanz. John D. Seeger is employed by Optum and has stock in UnitedHealth Group (Optum’s parent company). Participation in this project was supported through a research contract between Optum and Intercept. Gideon M. Hirschfield consults for Intercept, Advanz, Ipsen, Cymabay, HIghTide, Kowa, Pliant, Mirum, Gilead, GSK, and Escient. The remaining author has no conflicts to report.
Figures


References
-
- Frieden TR. Evidence for health decision making—Beyond randomized, controlled trials. N Engl J Med. 2017;377:465–475. - PubMed
-
- Code of Federal Regulations . Accelerated approval of new drugs for serious or life-threatening illnesses. CFR Title 21, Part 314, Subpart H., 1992.
-
- United States Food and Drug Adminisration. Submitting Documents Using Real-World Data and Real-World Evidence to FDA for Drug and Biological Products. Center for Drug Evaluation and Research; 2022.
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

