TRPM channels in human cancers: regulatory mechanism and therapeutic prospects
- PMID: 39633507
- PMCID: PMC11616203
- DOI: 10.1186/s40364-024-00699-2
TRPM channels in human cancers: regulatory mechanism and therapeutic prospects
Abstract
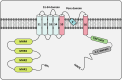
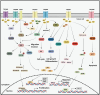
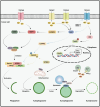
The transient receptor potential melastatin (TRPM) channel family has been previously implicated in various diseases, including those related to temperature sensing, cardiovascular health, and neurodegeneration. Nowadays, increasing evidence indicates that TRPM family members also play significant roles in various types of cancers, exhibiting both pro- and anti-tumorigenic functions. They are involved in tumor cell proliferation, survival, invasion, and metastasis, serving as potential diagnostic and prognostic biomarkers for cancer. This paper begins by describing the structure and physiological functions of the TRPM family members. It then outlines their roles in several common malignancies, including pancreatic, prostate, colorectal, breast, brain cancer, and melanoma. Subsequently, we focused on investigating the specific mechanisms by which TRPM family members are involved in tumorigenesis and development from both the tumor microenvironment (TME) and intracellular signaling. TRPM channels not only transmit signals from the TME to regulate tumor cell functions, but also mediate extracellular matrix remodeling, which is conducive to the malignant transformation of tumor cells. Importantly, TRPM channels depend on the regulation of the inflow of various ions in cells, and participate in key signaling pathways involved in tumor progression, such as Wnt/β-catenin, MAPK, PI3K/AKT, p53, and autophagy. Finally, we summarize the current strategies and challenges of targeting TRPM channels in tumor treatment, and discuss the feasibility of combining targeted TRPM channel drugs with cancer immunotherapy.
Keywords: Autophagy; Cancer progression; Signaling pathway; TRPM channels; Targeted tumor therapy; Tumor microenvironment.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Not applicable. Consent for publication: All the authors agree to publish the present work. Competing interests: The authors declare no competing interests.
Figures

Similar articles
-
The oncogenic roles of TRPM ion channels in cancer.J Cell Physiol. 2019 Sep;234(9):14556-14573. doi: 10.1002/jcp.28168. Epub 2019 Feb 2. J Cell Physiol. 2019. PMID: 30710353 Review.
-
The regulatory and modulatory roles of TRP family channels in malignant tumors and relevant therapeutic strategies.Acta Pharm Sin B. 2022 Apr;12(4):1761-1780. doi: 10.1016/j.apsb.2021.11.001. Epub 2021 Nov 5. Acta Pharm Sin B. 2022. PMID: 35847486 Free PMC article. Review.
-
On the modulation of TRPM channels: Current perspectives and anticancer therapeutic implications.Front Oncol. 2023 Feb 9;12:1065935. doi: 10.3389/fonc.2022.1065935. eCollection 2022. Front Oncol. 2023. PMID: 36844925 Free PMC article. Review.
-
Cell death modulation by transient receptor potential melastatin channels TRPM2 and TRPM7 and their underlying molecular mechanisms.Biochem Pharmacol. 2021 Aug;190:114664. doi: 10.1016/j.bcp.2021.114664. Epub 2021 Jun 25. Biochem Pharmacol. 2021. PMID: 34175300 Review.
-
A structural overview of the ion channels of the TRPM family.Cell Calcium. 2020 Jan;85:102111. doi: 10.1016/j.ceca.2019.102111. Epub 2019 Nov 24. Cell Calcium. 2020. PMID: 31812825 Free PMC article. Review.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

