Regioselective Dimerization of Methylcyclopentadiene inside Cucurbit[7]uril
- PMID: 39644128
- PMCID: PMC11855239
- DOI: 10.1002/chem.202403964
Regioselective Dimerization of Methylcyclopentadiene inside Cucurbit[7]uril
Abstract
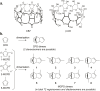
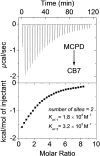
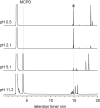
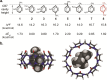
The molecular confinement within rigid macrocyclic receptors can trigger catalytic activity and steer the selectivity of organic reactions. In this work, the dimerization of methylcyclopentadiene (MCPD) isomers in the presence of cucurbit[7]uril (CB7) was found to display, besides a large rate acceleration, a striking regioselectivity in aqueous solution at pH 3, different from the products predominating in the absence of the supramolecular catalyst. Among the different possible regioisomers and diastereomers, the endo-3,7-dimethyl-3a,4,7,7a-tetrahydro-1H-4,7-methanoindene adduct was selectively formed, which is otherwise found only as a minor product in the dimerization of neat MCPD or in commercial dimeric mixtures. This product originates from the reaction of the heteroternary complex of 1-MCPD and 2-MCPD within CB7, in which the methyl groups are positioned in an "anti-diaxial" arrangement and point towards the open portals of the macrocycle, resulting in a preferred packing of the reacting cyclopentadiene rings. The selectivity of the dimerization of MCPD in the absence and presence of CB7 is supported by quantum-chemical calculations.
Keywords: Host-Guest Complexes; Macrocycles; Rate Acceleration; Regioisomers; Supramolecular Chemistry.
© 2024 The Author(s). Chemistry - A European Journal published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Raynal M., Ballester P., Vidal-Ferran A., van Leeuwen P. W. N. M., Chem. Soc. Rev. 2014, 43, 1734–1787. - PubMed
-
- Takashima Y., Harada A., in Encyclopedia of Polymeric Nanomaterials, (Eds.: Kobayashi S., Müllen K.), Springer, Berlin, Heidelberg, 2015, pp. 2366–2372.
-
- Pemberton B. C., Raghunathan R., Volla S., Sivaguru J., Chem. Eur. J. 2012, 18, 12178–12190. - PubMed
-
- Hong C. M., Bergman R. G., Raymond K. N., Toste F. D., Acc. Chem. Res. 2018, 51, 2447–2455. - PubMed
-
- Gaeta C., La Manna P., De Rosa M., Soriente A., Talotta C., Ne P., Gaeta C., La Manna P., De Rosa M., Soriente A., Talotta C., Neri P., ChemCatChem 2021, 13, 1638–1658.
Grants and funding
LinkOut - more resources
Full Text Sources

