Microglia degrade Alzheimer's amyloid-beta deposits extracellularly via digestive exophagy
- PMID: 39644493
- PMCID: PMC11760508
- DOI: 10.1016/j.celrep.2024.115052
Microglia degrade Alzheimer's amyloid-beta deposits extracellularly via digestive exophagy
Abstract
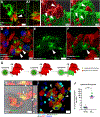
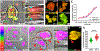
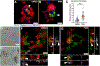
How microglia digest Alzheimer's fibrillar amyloid-beta (Aβ) plaques that are too large to be phagocytosed is not well understood. Here, we show that primary microglial cells create acidic extracellular compartments, lysosomal synapses, on model plaques and digest them with exocytosed lysosomal enzymes. This mechanism, called digestive exophagy, is confirmed by electron microscopy in 5xFAD mouse brains, which shows that a lysosomal enzyme, acid phosphatase, is secreted toward the plaques in structures resembling lysosomal synapses. Signaling studies demonstrate that the PI3K-AKT pathway modulates the formation of lysosomal synapses, as inhibition of PI3K1β or AKT1/2 reduces both lysosome exocytosis and actin polymerization, both required for the formation of the compartments. Finally, we show that small fibrils of Aβ previously internalized and trafficked to lysosomes are exocytosed toward large Aβ aggregates by microglia. Thus, the release of lysosomal contents during digestive exophagy may also contribute to the spread and growth of fibrillar Aβ.
Keywords: 5xFAD; Alzheimer’s disease; CP: Neuroscience; acid phosphatase; amyloid-beta; digestive exophagy; electron microscopy; extracellular degradation; lysosomal pH; lysosome; microglia.
Copyright © 2024 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The chemical synthesis and uses of the pH-sensitive probe ApHID have been included and described in a pending patent application, for which S.S.-D. and F.R.M. are co-authors.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

