The intersection of the HER2-low subtype with endocrine resistance: the role of interconnected signaling pathways
- PMID: 39650068
- PMCID: PMC11621065
- DOI: 10.3389/fonc.2024.1461190
The intersection of the HER2-low subtype with endocrine resistance: the role of interconnected signaling pathways
Abstract
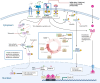
Since its introduction in the 1970s, endocrine therapy that targets the estrogen receptor alpha (ERα) signaling pathway has had tremendous success in the clinic in estrogen receptor positive (ER+) breast cancer. However, resistance to endocrine therapy eventually develops in virtually all patients with metastatic disease. Endocrine resistance is a primary unaddressed medical need for ER+ metastatic breast cancer patients. It has been shown that tumors become resistant through various mechanisms, converging on the acquisition of genetic alterations of ER, components of the MAP kinase pathway, or transcription factors (TFs). For instance, mutations in the human epidermal growth factor receptor-2 (HER2) lead to complete resistance to all current endocrine therapies including aromatase inhibitors, selective estrogen receptor modulators, and selective estrogen receptor degraders, as well as cross-resistance to CDK4/6 inhibitors (CDK4/6is). Emerging evidence points to an intriguing connection between endocrine-resistant tumors and the HER2-low subtype. Specifically, recent studies and our analysis of a publicly available breast cancer dataset both indicate that metastatic ER+ breast cancer with endocrine resistance conferred through acquired genetic alterations can often be classified as HER2-low rather than HER2-0/HER2-negative. Limited data suggest that acquired endocrine resistance can also be accompanied by a subtype switch. Therefore, we suggest that there is an underappreciated association between the HER2-low subtype and endocrine resistance. In this perspective piece, we explore the evidence linking the HER2-low subtype with the various pathways to endocrine resistance and suggest that there are signaling networks in HER2-low tumors that intersect endocrine resistance and can be effectively targeted.
Keywords: ER-HER2 crosstalk; HER2 mutations; combination therapy; endocrine resistance; subtype switch.
Copyright © 2024 Yayli, Tokofsky and Nayar.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Cristofanilli M, Turner NC, Bondarenko I, Ro J, Im SA, Masuda N, et al. Fulvestrant plus palbociclib versus fulvestrant plus placebo for treatment of hormone-receptor-positive, HER2-negative metastatic breast cancer that progressed on previous endocrine therapy (PALOMA-3): final analysis of the multicentre, double-blind, phase 3 randomised controlled trial. Lancet Oncol. (2016) 17:425–39. doi: 10.1016/S1470-2045(15)00613-0 - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

