Enhanced hepatitis B virus-specific immunity by combining neutralizing antibody therapy and DNA vaccination in a murine model of chronic hepatitis B virus infection
- PMID: 39652775
- PMCID: PMC12266798
- DOI: 10.1097/HEP.0000000000001179
Enhanced hepatitis B virus-specific immunity by combining neutralizing antibody therapy and DNA vaccination in a murine model of chronic hepatitis B virus infection
Abstract
Background and aims: Successful treatment of chronic HBV infection remains a great challenge due to the difficulty in inducing efficient immune responses. Here, we investigated the therapeutic potential of DNA vaccination combined with a potent HBV broadly neutralizing antibody targeting the small surface viral antigen.
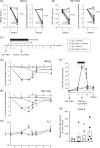
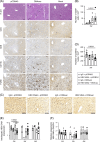
Approach and results: C57BL/6 mice were transduced with adeno-associated virus-HBV and were treated twice a week with HBV broadly neutralizing antibodies for 5 weeks. A DNA-based vaccine encoding the HBV core, envelope, and polymerase proteins was administered once to mice 3 weeks after initiating antibody therapy. The antiviral effects and antigen-specific immune responses were evaluated before and for 8 weeks after therapeutic vaccination. Vaccine administration with or without antibody treatment induced the development of functional HBV-specific CD8+ T cells and envelope-specific resident memory T cells in the liver. The combination of antibody treatment and DNA vaccination enhanced the recruitment of B and CD8+ T lymphocytes into the liver of HBV-carrier mice 2 weeks after vaccination. However, although still detectable 2 months after vaccination, HBV-specific CD8+ T cells showed an exhausted phenotype, suggesting that they are dysfunctional. In contrast, more effective control of antigenemia was observed following combination therapy, which was associated with the presence of HBs-specific memory B cells.
Conclusions: Although the combination therapy did not result in a functional cure, our findings indicate it produced additive effects on the development of HBV-specific T cells in the liver immediately following treatment, offering a better insight into the mechanisms underlying hepatic tolerance.
Keywords: T-cell response; antibody; chronic HBV infection; immune therapy; therapeutic vaccination.
Copyright © 2024 The Author(s). Published by Wolters Kluwer Health, Inc.
Conflict of interest statement
Malika Ait-Goughoulte is employed by Hoffmann La Roche. Hugo Mouquet and Maryline Bourgine report grants from Institut Roche during the conduct of the study. Maxime Beretta, Malika Ait-Goughoulte, Nadège Pelletier, Hugo Mouquet and Maryline Bourgine reported a patent to anti-HBV antibodies and methods of use, WO/2021/249990. The remaining authors have no conflicts to report.
Figures






Similar articles
-
High antigen burden drives CD8+ T cell dysfunction in a mouse model of chronic hepatitis B virus infection.J Virol. 2025 Jul 22;99(7):e0071125. doi: 10.1128/jvi.00711-25. Epub 2025 Jun 12. J Virol. 2025. PMID: 40503880 Free PMC article.
-
The Impact of Hepatitis B Surface Antigen Reduction via Small Interfering RNA Treatment on Natural and Vaccine (BRII-179)-Induced Hepatitis B Virus-Specific Humoral and Cellular Immune Responses.Gastroenterology. 2025 Jul;169(1):136-149. doi: 10.1053/j.gastro.2025.02.016. Epub 2025 Mar 3. Gastroenterology. 2025. PMID: 40043858 Clinical Trial.
-
Phase Ib/IIa randomized study of heterologous ChAdOx1-HBV/MVA-HBV therapeutic vaccination (VTP-300) as monotherapy and combined with low-dose nivolumab in virally-suppressed patients with CHB.J Hepatol. 2024 Dec;81(6):949-959. doi: 10.1016/j.jhep.2024.06.027. Epub 2024 Jul 6. J Hepatol. 2024. PMID: 38972484 Clinical Trial.
-
Hepatitis B immunoglobulin during pregnancy for prevention of mother-to-child transmission of hepatitis B virus.Cochrane Database Syst Rev. 2017 Feb 11;2(2):CD008545. doi: 10.1002/14651858.CD008545.pub2. Cochrane Database Syst Rev. 2017. PMID: 28188612 Free PMC article.
-
Combined DTP-HBV-HIB vaccine versus separately administered DTP-HBV and HIB vaccines for primary prevention of diphtheria, tetanus, pertussis, hepatitis B and Haemophilus influenzae B (HIB).Cochrane Database Syst Rev. 2012 Apr 18;2012(4):CD005530. doi: 10.1002/14651858.CD005530.pub3. Cochrane Database Syst Rev. 2012. PMID: 22513932 Free PMC article.
References
-
- Polaris Observatory C . Global prevalence, treatment, and prevention of hepatitis B virus infection in 2016: A modelling study. Lancet Gastroenterol Hepatol. 2018;3:383–403. - PubMed
-
- Naggie S, Lok AS. New therapeutics for hepatitis B: The road to cure. Annu Rev Med. 2021;72:93–105. - PubMed
-
- Hadziyannis SJ, Sevastianos V, Rapti I, Vassilopoulos D, Hadziyannis E. Sustained responses and loss of HBsAg in HBeAg-negative patients with chronic hepatitis B who stop long-term treatment with adefovir. Gastroenterology. 2012;143:629–636 e621. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

