Compensatory adaptation of parallel motor pathways promotes skilled forelimb recovery after spinal cord injury
- PMID: 39654633
- PMCID: PMC11626773
- DOI: 10.1016/j.isci.2024.111371
Compensatory adaptation of parallel motor pathways promotes skilled forelimb recovery after spinal cord injury
Abstract
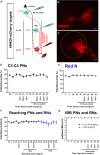
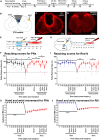
Skilled forelimb patterning is regulated by the corticospinal tract (CST) with support from brainstem regions. When the CST is lesioned, there is a loss of forelimb function; however, if indirect pathways remain intact, rehabilitative training can facilitate recovery. Following spinal cord injury, rehabilitation is thought to enhance the reorganization and plasticity of spared supraspinal-propriospinal circuits, aiding functional recovery. This study focused on the roles of cervical propriospinal interneurons (PNs) and rubrospinal neurons (RNs) in the recovery of reaching and grasping behaviors in rats with bilateral lesions of the CST and dorsal columns at C5. The lesions resulted in a 50% decrease in pellet retrieval, which normalized over four weeks of training. Silencing PNs or RNs after recovery resulted in reduced retrieval success. Notably, silencing both pathways corresponded to greater functional loss, underscoring their parallel contributions to recovery, alongside evidence of CST fiber sprouting in the spinal cord and red nucleus.
Keywords: Biological sciences; Natural sciences; Neuroscience; Systems neuroscience.
© 2024 The Authors. Published by Elsevier Inc.
Conflict of interest statement
The authors declare no competing interests.
Figures






References
-
- Lemon R.N. Descending pathways in motor control. Annu. Rev. Neurosci. 2008;31:195–218. - PubMed
-
- Han Q., Cao C., Ding Y., So K.F., Wu W., Qu Y., Zhou L. Plasticity of motor network and function in the absence of corticospinal projection. Exp. Neurol. 2015;267:194–208. - PubMed
-
- Kanagal S.G., Muir G.D. Bilateral dorsal funicular lesions alter sensorimotor behaviour in rats. Exp. Neurol. 2007;205:513–524. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

