The Molecular Mechanism Investigation of HBP-A Slows Down Meniscus Hypertrophy and Mineralisation by the Damage Mechanical Model
- PMID: 39656450
- PMCID: PMC11629809
- DOI: 10.1111/jcmm.70271
The Molecular Mechanism Investigation of HBP-A Slows Down Meniscus Hypertrophy and Mineralisation by the Damage Mechanical Model
Abstract
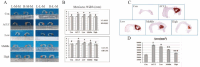
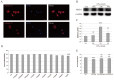
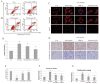
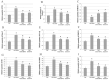
HBP-A is the main active component of a traditional Chinese medicine Huaizhen Yanggan Capsule, for the remarkable treatment of knee osteoarthritis (KOA). This study aimed to elucidate the ameliorative effect of HBP-A on meniscus hypertrophy and mineralisation in KOA and the molecular mechanism of its action. An Hartley guinea pig model of KOA that underwent anterior cruciate ligament transection (ACLT) and a model of rat primary meniscus fibrochondrocytes (PMFs) were used to investigate the ameliorative effect of HBP-A on meniscal hypertrophy and calcification and its signal transduction mechanism of action. The results show that Guinea pig's meniscus width, as well as the area of meniscus calcification and meniscus and articular cartilage injury score, were significantly reduced in the HBP-A intervention group compared to the ACLT group. The expression levels of mtrix metalloproteinase 13 (MMP13), runt-related transcription factor 2 (Runx2), Indian hedgehog (Ihh), alkaline phosphatase (ALP), and ankylosis homologue (ANKH) at the protein and gene level significantly decreased in the HBP-A intervention group compared to the ACLT group. In vitro study, apoptosis, hypertrophy, and calcification of rat PMFs after 10% stretch force were significantly improved with HBP-A intervention. Western blot and RT-qPCR showed that hypertrophy, calcification, and p38 MAPK signalling pathway-related markers of PMFs were incredibly depressed in the HBP-A intervention group compared to the 10% stretch force group. In conclusion, HBP-A can slow down meniscus hypertrophy and mineralisation induced by abnormal mechanical loading, and its mechanism of action may be through the p38-MAPK signalling pathway.
Keywords: HBP‐A; KOA; abnormal mechanical damage; hypertrophy and mineralisation; meniscus; p38‐MAPK signalling pathway.
© 2024 The Author(s). Journal of Cellular and Molecular Medicine published by Foundation for Cellular and Molecular Medicine and John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
Abnormal Mechanical Loading Induces Cartilage Degeneration by Accelerating Meniscus Hypertrophy and Mineralization After ACL Injuries In Vivo.Am J Sports Med. 2016 Mar;44(3):652-63. doi: 10.1177/0363546515621285. Epub 2016 Jan 20. Am J Sports Med. 2016. PMID: 26792705 Free PMC article.
-
Hesperetin Attenuates T-2 Toxin-Induced Chondrocyte Injury by Inhibiting the p38 MAPK Signaling Pathway.Nutrients. 2024 Sep 14;16(18):3107. doi: 10.3390/nu16183107. Nutrients. 2024. PMID: 39339707 Free PMC article.
-
[Effect of HBP-A on meniscal injury and pathological hypertrophy and calcification of the meniscus].Nan Fang Yi Ke Da Xue Xue Bao. 2016 Apr 20;37(4):431-437. doi: 10.3969/j.issn.1673-4254.2017.04.02. Nan Fang Yi Ke Da Xue Xue Bao. 2016. PMID: 28446392 Free PMC article. Chinese.
-
Adenovirus-mediated transduction with Histone Deacetylase 4 ameliorates disease progression in an osteoarthritis rat model.Int Immunopharmacol. 2019 Oct;75:105752. doi: 10.1016/j.intimp.2019.105752. Epub 2019 Jul 13. Int Immunopharmacol. 2019. PMID: 31310910
-
Inhibition against p38/MEF2C pathway by Pamapimod protects osteoarthritis chondrocytes hypertrophy.Panminerva Med. 2024 Dec;66(4):365-371. doi: 10.23736/S0031-0808.20.04170-1. Epub 2020 Dec 2. Panminerva Med. 2024. PMID: 33263251
Cited by
-
HBP-A Attenuates Knee Osteoarthritis Progression via MLK3/P38/HDAC4 Axis-Mediated Dual Protection of Articular Cartilage and Quadriceps.J Cell Mol Med. 2025 May;29(9):e70577. doi: 10.1111/jcmm.70577. J Cell Mol Med. 2025. PMID: 40318007 Free PMC article.
References
-
- Herrero‐Beaumont G., Roman‐Blas J. A., Bruyère O., et al., “Clinical Settings in Knee Osteoarthritis: Pathophysiology Guides Treatment,” Maturitas 96 (2017): 54–57. - PubMed
-
- Richter M., Trzeciak T., Rybka J. D., et al., “Correlations Between Serum Adipocytokine Concentrations, Disease Stage, Radiological Status and Total Body Fat Content in the Patients With Primary Knee Osteoarthritis,” International Orthopaedics 41, no. 5 (2017): 983–989. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

