Satiety Hormone LEAP2 After Low-Calorie Diet With/Without Endobarrier Insertion in Obesity and Type 2 Diabetes Mellitus
- PMID: 39659543
- PMCID: PMC11631353
- DOI: 10.1210/jendso/bvae214
Satiety Hormone LEAP2 After Low-Calorie Diet With/Without Endobarrier Insertion in Obesity and Type 2 Diabetes Mellitus
Abstract
Context: The liver/foregut satiety hormone liver-expressed antimicrobial peptide 2 (LEAP2) is an inverse agonist at the acyl ghrelin receptor (GHSR), increasing after food intake and decreasing after bariatric surgery and short-term nonsurgical weight loss, but effects of long-term dietary weight loss are unknown.
Objective: The objective of this study was to examine and compare the effects of these interventions on fasting and postprandial plasma LEAP2 and investigate potential metabolic mediators of changes in plasma LEAP2.
Methods: Plasma LEAP2 was measured in a previously published 2-year trial comparing standard medical management (SMM) (including 600-kcal/day deficit) with duodenal-jejunal bypass liner (DJBL, Endobarrier) insertion (explanted after 1 year) in adults with obesity and inadequately controlled type 2 diabetes mellitus.
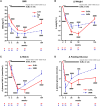
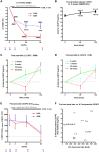
Results: In the SMM group (n = 25-37), weight decreased by 4.3%, 8.1%, 7.8%, and 6.4% at 2, 26, 50, and 104 weeks and fasting plasma LEAP2 decreased from baseline mean ± SD 15.3 ± 0.9 ng/mL by 1.7, 3.8, 2.1, and 2.0 ng/mL, respectively. Absolute/decreases in fasting plasma LEAP2 positively correlated with absolute/decreases in body mass index, glycated hemoglobin A1c, fasting plasma glucose, serum insulin, homeostatic model assessment for insulin resistance, and serum triglycerides. Despite greater weight loss in the DJBL group (n = 23-30) at 26 to 50 weeks (10.4%-11.4%), the decrease in fasting plasma LEAP2 was delayed and attenuated (vs SMM), which may contribute to greater weight loss by attenuating GHSR signaling. Plasma LEAP2 did not increase with weight regain from 50 to 104 weeks after DJBL explant, suggesting a new set point with weight loss maintenance. Increases in plasma LEAP2 after a 600-kcal meal (10.8%-16.1% at 1-2 hours) were unaffected by weight loss, improved glucose metabolism, or DJBL insertion (n = 9-25), suggesting liver rather than duodenum/jejunum may be the primary source of postprandial LEAP2 secretion.
Conclusion: These findings add to our understanding of the regulation and potential physiological role of plasma LEAP2.
Keywords: LEAP2; appetite; duodenal-jejunal bypass liner; glucose; insulin; obesity; weight loss.
© The Author(s) 2024. Published by Oxford University Press on behalf of the Endocrine Society.
Figures



References
-
- WHO . World health statistics 2022: monitoring health for the SDGs, sustainable development goals. 2022; Available from: https://www.who.int/publications/i/item/9789240051157
-
- Perdomo CM, Cohen RV, Sumithran P, Clement K, Fruhbeck G. Contemporary medical, device, and surgical therapies for obesity in adults. Lancet. 2023;401(10382):1116‐1130. - PubMed
-
- Korbonits M, Goldstone AP, Gueorguiev M, Grossman AB. Ghrelin—a hormone with multiple functions. Front Neuroendocrinol. 2004;25:27‐68. - PubMed
-
- Kojima M, Kangawa K. Drug insight: the functions of ghrelin and its potential as a multitherapeutic hormone. Nat Clin Pract Endocrinol Metab. 2006;2(2):80‐88. - PubMed
LinkOut - more resources
Full Text Sources
