Hypersensitive intercellular responses of endometrial stromal cells drive invasion in endometriosis
- PMID: 39660704
- PMCID: PMC11729374
- DOI: 10.7554/eLife.94778
Hypersensitive intercellular responses of endometrial stromal cells drive invasion in endometriosis
Abstract
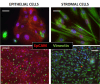
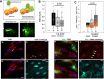
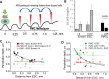
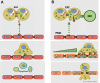
Endometriosis is a debilitating disease affecting 190 million women worldwide and the greatest single contributor to infertility. The most broadly accepted etiology is that uterine endometrial cells retrogradely enter the peritoneum during menses, and implant and form invasive lesions in a process analogous to cancer metastasis. However, over 90% of women suffer retrograde menstruation, but only 10% develop endometriosis, and debate continues as to whether the underlying defect is endometrial or peritoneal. Processes implicated in invasion include: enhanced motility; adhesion to, and formation of gap junctions with, the target tissue. Endometrial stromal (ESCs) from 22 endometriosis patients at different disease stages show much greater invasiveness across mesothelial (or endothelial) monolayers than ESCs from 22 control subjects, which is further enhanced by the presence of EECs. This is due to the enhanced responsiveness of endometriosis ESCs to the mesothelium, which induces migration and gap junction coupling. ESC-PMC gap junction coupling is shown to be required for invasion, while coupling between PMCs enhances mesothelial barrier breakdown.
Keywords: cell biology; endometriosis; gap junctions; human; invasion; motility.
© 2024, Chen, Chavez et al.
Conflict of interest statement
CC, JC, RK, VG, AP, AJ, SP, MH, RR, MG, PO No competing interests declared, NK, BN co-founder and shareholder of Hear Biotech Inc, developing a diagnostic for endometriosis in part based on these findings
Figures






Update of
References
-
- Acosta Go V-A, Chavez J, Robinson RD, Galang MA, Kumar RS, Nicholson B. Investigating the role of peritoneal mesothelial cells in endometriosis lesion formation. Fertility and Sterility. 2023;120:e318. doi: 10.1016/j.fertnstert.2023.08.916. - DOI
-
- Beahm DL, Oshima A, Gaietta GM, Hand GM, Smock AE, Zucker SN, Toloue MM, Chandrasekhar A, Nicholson BJ, Sosinsky GE. Mutation of a conserved threonine in the third transmembrane helix of alpha- and beta-connexins creates a dominant-negative closed gap junction channel. The Journal of Biological Chemistry. 2006;281:7994–8009. doi: 10.1074/jbc.M506533200. - DOI - PubMed
-
- Björk E, Israelsson P, Nagaev I, Nagaeva O, Lundin E, Ottander U, Mincheva-Nilsson L. Endometriotic tissue-derived exosomes downregulate NKG2D-mediated cytotoxicity and promote apoptosis: mechanisms for survival of ectopic endometrial tissue in endometriosis. Journal of Immunology. 2024;213:567–576. doi: 10.4049/jimmunol.2300781. - DOI - PMC - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

