Mayo Genetic Risk Models for Newly Diagnosed Acute Myeloid Leukemia Treated With Venetoclax + Hypomethylating Agent
- PMID: 39671248
- PMCID: PMC11705209
- DOI: 10.1002/ajh.27564
Mayo Genetic Risk Models for Newly Diagnosed Acute Myeloid Leukemia Treated With Venetoclax + Hypomethylating Agent
Abstract
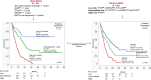
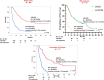
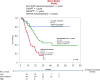
Patients with newly diagnosed acute myeloid leukemia (ND-AML) derive variable survival benefit from venetoclax + hypomethylating agent (Ven-HMA) therapy. The primary objective in the current study was to develop genetic risk models that are predictive of survival and are applicable at the time of diagnosis and after establishing treatment response. Among 400 ND-AML patients treated with Ven-HMA at the Mayo Clinic, 247 (62%) achieved complete remission with (CR) or without (CRi) count recovery. Multivariable analysis-derived hazard ratios (HR), including 1.8 for European LeukemiaNet (ELN) adverse karyotype, 4.7 for KMT2Ar, 1.7 for TP53 MUT, 2.6 for KRAS MUT, and 2.1 for IDH2 WT were applied to develop an HR-weighted risk model: low, intermediate, and high; respective median survival censored for allogeneic stem cell transplant (ASCT) (3-year survival) were "not reached" (67%), 19.1 (33%), and 7.1 months (0%). In patients achieving CR/CRi, adverse karyotype, KMT2Ar, KRAS MUT, IDH2 WT predicted inferior survival, allowing for a complementary response-stratified risk model. The model was externally validated and was shown to be superior to the ELN 2024 risk model (AIC 179 vs. 195 and AUC 0.77 vs. 0.69). Survival was inferior with failure to achieve CR/CRi or not receiving ASCT; 3-year survival for high-risk with or without ASCT was 42% versus 0% (p < 0.01); intermediate 72% versus 43% (p = 0.06); and low-risk 88% versus 78% (p = 0.53). The Mayo genetic risk models offer pre-treatment and response-based prognostic tools for ND-AML treated with Ven-HMA. The current study underscores the prognostically indispensable role of achieving CR/CRi and ASCT.
Keywords: karyotype; mutations; remission; survival; venetoclax.
© 2024 The Author(s). American Journal of Hematology published by Wiley Periodicals LLC.
Conflict of interest statement
M.R.L: Research support from Abbvie, Astellas, Amgen, Actinium, Pluristem, Sanofi. Speaker's Bureau for Amgen, Beigene. Data Safety Monitoring Committee for Biosight. N.G.: Advisory Board for DISC medicine and Agios. M.S.: Research funding to the institution from Astellas, Celgene, and Marker Therapeutics. M.M.P.: Member of the board of directors or advisory committees of Stemline Therapeutics and received research funding from Kura Oncology. A.A.P.: Research funding from Pfizer, Kronos Bio, Sumitomo; honoraria from AbbVie, Sobi, Bristol Myers Squibb. ED: Honoraria from AbbVie. C.D.: Consultant/Advisory Boards: Abbvie, AstraZeneca, Astellas, BMS, Genentech, GenMab, GSK, Notable Labs, Rigel, Ryvu, Schrodinger, Servier. C.D.: is supported by the LLS Scholar in Clinical Research Award. T.K.: Grant support: BMS, Abbvie, Amgen, Ascentage Pharma Group, Astellas Pharma, DrenBio, Astex, AstraZeneca, BMS, Celgene, Incyte, Cellenkos, Cyclacel, Delta‐Fly pharma, Genentech, Genfleet, Glycomimetics, Iterion, Janssen, Jazz Pharmaceuticals, Pfizer, Pulmotect, Regeneron, SELLAS. Consulting fees: AbbVie, Agios, Daiichi Sankyo, Genentech, Genzyme, Jazz Pharmaceuticals, Liberum, Novartis, Pfizer, PinotBio, Pukmotect, Sanofi‐Aventis, Servier. Payment or honoraria: AbbVie, Agios, Daiichi Sankyo, DAVA Oncology, Delta‐Fly, DrenBio, Genentech, Genfleet, Genzyme, Jazz Pharmaceuticals, Liberum, Novartis, Pfizer, Rigel, Sanofi‐Aventis, SELLAS, Servier. The remaining authors declare no competing financial interests.
Figures

References
-
- DiNardo C. D., Pratz K. W., Letai A., et al., “Safety and Preliminary Efficacy of Venetoclax With Decitabine or Azacitidine in Elderly Patients With Previously Untreated Acute Myeloid Leukaemia: A Non‐randomised, Open‐Label, Phase 1b Study,” Lancet Oncology 19, no. 2 (2018): 216–228. - PubMed
-
- DiNardo C. D., Jonas B. A., Pullarkat V., et al., “Azacitidine and Venetoclax in Previously Untreated Acute Myeloid Leukemia,” New England Journal of Medicine 383, no. 7 (2020): 617–629. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

