Canagliflozin treatment prevents follicular exhaustion and attenuates hallmarks of ovarian aging in genetically heterogenous mice
- PMID: 39672978
- PMCID: PMC12181533
- DOI: 10.1007/s11357-024-01465-w
Canagliflozin treatment prevents follicular exhaustion and attenuates hallmarks of ovarian aging in genetically heterogenous mice
Abstract
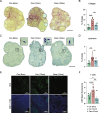
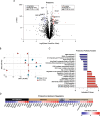
Ovarian aging is characterized by declines in follicular reserve and the emergence of mitochondrial dysfunction, reactive oxygen species production, inflammation, and fibrosis, which eventually results in menopause. Menopause is associated with increased systemic aging and the development of numerous comorbidities; therefore, the attenuation of ovarian aging could also delay systemic aging processes in women. Recent work has established that the anti-diabetic drug Canagliflozin (Cana), a sodium-glucose transporter 2 inhibitor, elicits benefits on aging-related outcomes, likely through the modulation of nutrient-sensing pathways and metabolic homeostasis. Given that nutrient-sensing pathways play a critical role in controlling primordial follicle activation, we sought to determine if chronic Cana administration would delay ovarian aging and curtail the emergence of pathological hallmarks associated with reproductive senescence. We found that mice receiving Cana maintained their ovarian reserve through 12 months of age, which was associated with declines in primordial follicles FoxO3a phosphorylation, a marker of activation, when compared to the age-matched controls. Furthermore, Cana treatment led to decreased collagen, lipofuscin, and T cell accumulation at 12 months of age. Whole ovary transcriptomic and proteomic analyses revealed subtle improvements, predominantly in mitochondrial function and the regulation of cellular proliferation. Pathway analyses of the transcriptomic data revealed a downregulation in cell proliferation and mitochondrial dysfunction signatures, with an upregulation of oxidative phosphorylation. Pathway analyses of the proteomic data revealed declines in signatures associated with PI3K/AKT activity and lymphocyte accumulation. Collectively, we demonstrate that Cana treatment can delay ovarian aging in mice and could potentially have efficacy for delaying ovarian aging in women.
Keywords: Fertility; Fibrosis; Glucose; Insulin; Ovary; SGLT2.
© 2024. The Author(s), under exclusive licence to American Aging Association.
Conflict of interest statement
Declarations. Disclaimer: The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. Conflict of interest: The authors declare no competing interests.
Figures




References
MeSH terms
Substances
Grants and funding
- R01 ES033171/ES/NIEHS NIH HHS/United States
- R56 ES034765/ES/NIEHS NIH HHS/United States
- P20 GM103447/GM/NIGMS NIH HHS/United States
- AG069742/AG/NIA NIH HHS/United States
- P30 AG050911/AG/NIA NIH HHS/United States
- GCRLE-4501/Global Consortium for Reproductive Longevity and Equality
- RF1 AG078170/AG/NIA NIH HHS/United States
- P30 DK020572/DK/NIDDK NIH HHS/United States
- P20 GM139763/GM/NIGMS NIH HHS/United States
- R24 GM137786/GM/NIGMS NIH HHS/United States
- GCRLE-0523/Global Consortium for Reproductive Longevity and Equality
- R01 AG069742/AG/NIA NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Research Materials

