T1 and T2 measurements of the neonatal brain at 7 T
- PMID: 39673110
- PMCID: PMC7617262
- DOI: 10.1002/mrm.30403
T1 and T2 measurements of the neonatal brain at 7 T
Abstract
Purpose: To determine the expected range of NMR relaxation times (T1 and T2) in the neonatal brain at 7 T.
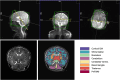
Methods: Data were acquired in a total of 40 examinations on infants in natural sleep. The cohort included 34 unique subjects with postmenstrual age range between 33 and 52 weeks and contained a mix of healthy individuals and those with clinical concerns. Single-slice T1 and T2 mapping protocols were used to provide measurements in white matter, cortex, cerebellum, and deep gray matter. Automatic image segmentation of a separate T2-weighted brain volume was used to define regions of interest for analysis.
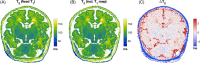
Results: Linear regression was used to estimate relaxation times at term equivalent age (40 weeks postmenstrual age). with 95% confidence intervals was measured to be 2933 [2893, 2972] ms in white matter; 2653 [2604, 2701] ms in cerebellum; and 2486 [2439, 2532] ms in basal ganglia. was estimated as 119 [116, 121] ms in white matter, 99 [96, 102] ms in cerebellum, and 90 [89, 92] ms in basal ganglia. Most tissue-relaxation times showed a significant negative correlation with postmenstrual age, with the strongest correlation seen in cerebellum.
Conclusions: We describe neonatal brain tissue and age-specific T1 and T2 relaxation values at 7 T. The presented values differ substantially from both adult values at 7 T and neonate values measured at lower field strengths, and will be essential for pulse-sequence optimization for neonatal studies.
Keywords: neonatal imaging; relaxation times; ultrahigh field.
© 2024 The Author(s). Magnetic Resonance in Medicine published by Wiley Periodicals LLC on behalf of International Society for Magnetic Resonance in Medicine.
Conflict of interest statement
Raphael Tomi‐Tricot is an employee of Siemens Healthcare Limited.
Figures


References
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

