Direct Interaction Between CD34+ Hematopoietic Stem Cells and Mesenchymal Stem Cells Reciprocally Preserves Stemness
- PMID: 39682159
- PMCID: PMC11640414
- DOI: 10.3390/cancers16233972
Direct Interaction Between CD34+ Hematopoietic Stem Cells and Mesenchymal Stem Cells Reciprocally Preserves Stemness
Abstract
Background/objectives: A specialized microenvironment in the bone marrow, composed of stromal cells including mesenchymal stem cells (MSCs), supports hematopoietic stem cell (HSC) self-renewal, and differentiation bands play an important role in leukemia development and progression. The reciprocal direct interaction between MSCs and CD34+ HSCs under physiological and pathological conditions is yet to be fully characterized.
Methods: Here, we established a direct co-culture model between MSCs and CD34+ HSCs or MSCs and acute myeloid leukemia cells (THP-1, Molm-13, and primary cells from patients) to study heterocellular communication.
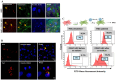
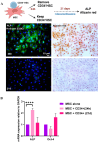
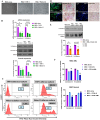
Results: Following MSCs-CD34+ HSCs co-culture, the expression of adhesion markers N-Cadherin and connexin 43 increased in both cell types, forming gap junction channels. Moreover, the clonogenic potential of CD34+ HSCs was increased. However, direct contact of acute myeloid leukemia cells with MSCs reduced the expression levels of connexin 43 and N-Cadherin in MSCs. The impairment in gap junction formation may potentially be due to a defect in the acute myeloid leukemia-derived MSCs. Interestingly, CD34+ HSCs and acute myeloid leukemia cell lines attenuated MSC osteoblastic differentiation upon prolonged direct cell-cell contact.
Conclusions: In conclusion, under physiological conditions, connexin 43 and N-Cadherin interaction preserves stemness of both CD34+ HSCs and MSCs, a process that is compromised in acute myeloid leukemia, pointing to the possible role of gap junctions in modulating stemness.
Keywords: CD34+ hematopoietic stem cells; N-Cadherin; acute myeloid leukemia; bone marrow; connexin 43; gap junction; heterocellular interaction; mesenchymal stem cells; microenvironment.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

