Frequency and Significance of Body Weight Loss During Immunochemotherapy in Patients with Advanced Non-Small Cell Lung Cancer
- PMID: 39682275
- PMCID: PMC11640719
- DOI: 10.3390/cancers16234089
Frequency and Significance of Body Weight Loss During Immunochemotherapy in Patients with Advanced Non-Small Cell Lung Cancer
Abstract
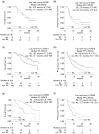
Background: Limited data are available on the frequency and significance of body weight loss during cancer therapy. This study investigated the frequency of patients who experienced body weight loss during immune checkpoint inhibitor (ICI) plus chemotherapy for advanced non-small cell lung cancer (NSCLC) and the impact of weight loss on treatment outcomes. Methods: Using the clinical data of 370 patients with NSCLC who received a combination of ICI and chemotherapy at 13 institutions, this study investigated the frequency of body weight loss > 5% during treatment and determined the impact of body weight loss on patient outcomes. Results: Of the 370 included patients, 141 (38.1%) lost more than 5% of their body weight during ICI plus chemotherapy (WL group). The 2-month landmark analysis showed that patients who experienced body weight loss of >5% during treatment had worse overall survival (OS) and progression-free survival (PFS) than those who did not (OS 14.0 and 31.1 months in the WL non-WL groups, respectively, p < 0.001; PFS 6.8 and 10.9 months in the WL non-WL groups, respectively, p = 0.002). Furthermore, a negative impact of body weight loss on survival was observed even in those who had obesity (body mass index [BMI] ≥ 25.0) at the start of therapy (OS 12.8 and 25.4 months in the WL non-WL groups, respectively, p < 0.001; PFS 5.7 and 10.7 months in the WL non-WL groups, respectively, p = 0.038). Conclusions: In conclusion, weight loss of >5% during ICI plus chemotherapy negatively influenced patient outcomes. Further and broader studies should investigate the role of nutritional status, specifically weight change and nutritional support, in responsiveness to ICI plus chemotherapy.
Keywords: body weight loss; chemotherapy; immune checkpoint inhibitors; non-small cell lung cancer.
Conflict of interest statement
Masataka Taoka received honoraria from AstraZeneca K.K. Eiki Ichihara has received honoraria from AstraZeneca K.K., Novartis Pharma K.K., Janssen Pharmaceutical K.K., Chugai Pharmaceutical Co., Ltd., Eli Lilly Japan K.K., Pfizer Japan Inc., Bristol-Myers Squibb K.K., ONO Pharmaceutical Co., Ltd., Takeda Pharmaceutical Company Limited, and Boehringer Ingelheim. Eiki Ichihara received research funding from Janssen Pharmaceutical K.K.; Pfizer Japan Inc.; Bristol-Myers Squibb K.K.; ONO Pharmaceutical Co., Ltd.; and Takeda Pharmaceutical Company, Ltd. Toshihide Yokoyama received honoraria from AstraZeneca K.K., Chugai Pharmaceutical Co., Ltd., Eli Lilly Japan K.K., Bristol-Myers Squibb K.K., ONO Pharmaceutical Co., Ltd., MSD K.K., and Nippon Kayaku Co., Ltd. Toshihide Yokoyama received grants from AstraZeneca K.K., Boehringer Ingelheim, Chugai Pharmaceutical Co., Ltd., Bristol-Myers Squibb K.K., MSD K.K., Daiichi Sankyo Co., Ltd., and Parexel International Inc. Tomoki Tamura received honoraria from AstraZeneca K.K., Chugai Pharmaceutical Co., Ltd., Kyowa Kirin Co., Ltd., and Boehringer Ingelheim. Hirohisa Kano received honoraria from Chugai Pharmaceutical Co., Ltd., MSD K.K., AstraZeneca K.K., Bristol-Myers Squibb K.K., and ONO Pharmaceutical Co., Ltd. Haruyuki Kawai has received honoraria from Glaxo Smith Kline, Meiji Seika Pharma Co., Ltd., Chugai Pharmaceutical Co., Ltd., Taiho Pharmaceutical Co., Ltd., MSD K.K., Nippon Kayaku Co., Ltd., AstraZeneca K.K., Bristol-Myers Squibb K.K., Boehringer Ingelheim, and Novartis Pharma K.K. Masaaki Inoue received honoraria from AstraZeneca K.K., Chugai Pharmaceutical Co., Ltd., MSD K.K., Bristol-Myers Squibb K.K., Daiichi-Sankyo Co., Ltd., and Nippon Kayaku Co., Ltd. Nobuaki Fujimoto received honoraria from ONO Pharmaceutical Co., Ltd., AstraZeneca K.K., Chugai Pharmaceutical Co., Ltd., Taiho Pharmaceutical Co., Ltd., MSD K.K., and Nippon Kayaku Co., Ltd. Nobuaki Fujimoto received consulting fees from AstraZeneca K.K. Katsuyuki Kiura has received honoraria from Merck Biopharma, Taiho Pharmaceutical Co., Ltd., Chugai Pharmaceutical Co., Ltd., Takeda Pharmaceutical Company Limited, Boehringer Ingelheim, Eli Lilly Japan K.K., Pfizer Japan Inc., and Bristol-Myers Squibb K.K. Katsuyuki Kiura has received grants from Teijin Pharma Limited, Shionogi Pharmaceutical Co., Ltd., Boehringer Ingelheim, Nippon Kayaku Co., Ltd., Taiho Pharmaceutical Co., Ltd., and Chugai Pharmaceutical Co., Ltd. Katsuyuki Kiura received consulting fees from Nippon Kayaku Co., Ltd. and Nipro K. K. Yoshinobu Maeda has received honoraria from Asahi Kasei Pharma Corporation, Eisai Co., Ltd., Otsuka Pharmaceutical Co., Ltd., Kyowa Kirin Co., Ltd., KYORIN Pharmaceutical Co., Ltd., Taiho Pharmaceutical Co., Ltd., Takeda Pharmaceutical Company Limited, Chugai Pharmaceutical Co., Ltd., Teijin Pharma Limited, Japan Blood Products Organization, NIPPON KAYAKU Co., Ltd., Nippon Shinyaku Co., Ltd., Mallinckrodt Pharma K.K., and REGiMMUNE Co., Ltd. Yoshinobu Maeda received grants from Asahi Kasei Pharma Corporation, Astellas Pharma Inc., AstraZeneca K.K., Amgen K.K., AbbVie GK, Eisai Co., Ltd., Viatris Inc., Otsuka Pharmaceutical Co., Ltd., ONO Pharmaceutical Co., Ltd., KYORIN Pharmaceutical Co., Ltd., KISSEI Pharmaceutical Co., Ltd., Kyowa Kirin Co., Ltd., Gilead Sciences, Inc., KONICA MINOLTA, Inc., Sanofi K.K., Sumitomo Dainippon Pharma Co., Ltd., JCR Pharmaceuticals Co., Ltd., Celgene Corporation, CSL Behring K.K., Daiichi Sankyo Co., Ltd., Takeda Pharmaceutical Company Limited, TERUMO Corporation, Chugai Pharmaceutical Co., Ltd., Nippon Shinyaku Co., Ltd., Bayer Yakuhin, Ltd., Bristol-Myers Squibb K.K., Novartis Pharma K.K., Pfizer Japan Inc., Pharma Essentia Corp., Mundipharma K.K., Human Life CORD Japan Inc., Meiji Seika Pharma Co., Ltd., Medical Review Co., Ltd., Janssen Pharmaceutical K.K., and Yakult Honsha Co., Ltd. Yoshinobu Maeda received grants from research funds under contracts with Chugai Pharmaceutical Co., Ltd. and Nippon Shinyaku Co., Ltd. Katsuyuki Hotta received honoraria from AstraZeneca K.K., Chugai Pharmaceutical Co., Ltd., Eli Lilly Japan K.K., Bristol-Myers Squibb K.K., ONO Pharmaceutical Co., Ltd., MSD K.K., Nippon Kayaku Co., Ltd., Amgen K.K., Taiho Pharmaceutical Co., Ltd., Merck, and Boehringer Ingelheim. Katsuyuki Hotta received research funding from AstraZeneca K.K., Chugai Pharmaceutical Co., Ltd., Bristol-Myers Squibb K.K., ONO Pharmaceutical Co., Ltd., Takeda Pharmaceutical Company Limited, MSD K.K., Eli Lilly Japan K.K., and AbbVie GK. The authors have no conflicts of interest to declare.
Figures

References
-
- Socinski M.A., Jotte R.M., Cappuzzo F., Orlandi F., Stroyakovskiy D., Nogami N., Rodríguez-Abreu D., Moro-Sibilot D., Thomas C.A., Barlesi F., et al. Atezolizumab for First-Line Treatment of Metastatic Nonsquamous NSCLC. N. Engl. J. Med. 2018;378:2288–2301. doi: 10.1056/NEJMoa1716948. - DOI - PubMed
-
- West H., McCleod M., Hussein M., Morabito A., Rittmeyer A., Conter H.J., Kopp H.G., Daniel D., McCune S., Mekhail T., et al. Atezolizumab in Combination with Carboplatin Plus Nab-Paclitaxel Chemotherapy Compared with Chemotherapy Alone as First-Line Treatment for Metastatic Non-Squamous Non-Small-Cell Lung Cancer (IMpower130): A Multicentre, Randomised, Open-Label, Phase 3 Trial. Lancet Oncol. 2019;20:924–937. doi: 10.1016/S1470-2045(19)30167-6. - DOI - PubMed
-
- Paz-Ares L., Ciuleanu T.E., Cobo M., Schenker M., Zurawski B., Menezes J., Richardet E., Bennouna J., Felip E., Juan-Vidal O., et al. First-Line Nivolumab Plus Ipilimumab Combined with Two Cycles of Chemotherapy in Patients with Non-Small-Cell Lung Cancer (CheckMate 9LA): An International, Randomised, Open-Label, Phase 3 Trial. Lancet Oncol. 2021;22:198–211. doi: 10.1016/S1470-2045(20)30641-0. - DOI - PubMed
LinkOut - more resources
Full Text Sources

