Exploration of Alkyne-Based Multilayered 3D Polymers and Oligomers: Subtle Aggregation-Induced Emission, Chromium(VI) Ion Detection, and Chiral Properties Characterization
- PMID: 39683800
- PMCID: PMC11643666
- DOI: 10.3390/molecules29235641
Exploration of Alkyne-Based Multilayered 3D Polymers and Oligomers: Subtle Aggregation-Induced Emission, Chromium(VI) Ion Detection, and Chiral Properties Characterization
Abstract
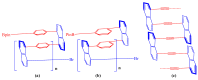
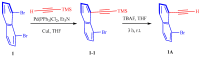
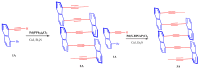
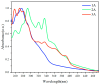
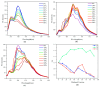
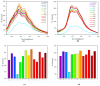
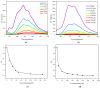
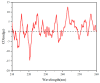
This study investigates the synthesis and characterization of alkyne-based multilayered three-dimensional (3D) polymers, which exhibit a subtle aggregation-induced emission (AIE) phenomenon. The polymers demonstrate significant potential as fluorescent probes for the selective detection of chromium (VI) ions (Cr6⁺), showcasing their utility in environmental sensing applications. Additionally, the circular dichroism (CD) spectra reveal a pronounced cotton effect, indicative of chiral properties, while scanning electron microscopy (SEM) and dynamic light scatting (DLS) analysis reveal a distinctive rock-like surface morphology and Cr6+ sensitive anti-aggregation. These findings highlight the multifunctional capabilities of alkyne-based multilayered 3D polymers, suggesting their applicability in both fluorescence-based sensing and materials science. The insights gained from this research contribute to the development of advanced materials with tailored optical properties for environmental monitoring and other practical applications.
Keywords: aggregation-induced emission; chiral properties characterization; chromium (VI) ion detection; multilayered 3D polymer.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper. Data availability: Data will be made available upon request.
Figures

Similar articles
-
Synthesis and Characterization of Multilayer 3D Chiral Polymers with Enhanced Optical Properties.Molecules. 2025 Mar 31;30(7):1567. doi: 10.3390/molecules30071567. Molecules. 2025. PMID: 40286155 Free PMC article.
-
Aggregation-induced emission in novel multilayered 3D polymers: development of a fluorescent probe for selective metal ion detection.RSC Adv. 2025 Jan 23;15(3):2242-2249. doi: 10.1039/d4ra08548e. eCollection 2025 Jan 16. RSC Adv. 2025. PMID: 39850082 Free PMC article.
-
Characterization of Chiral Thiophene Multilayer 3D Polymers with AIE Properties for Environmental Monitoring of Chromium Ions.Macromol Rapid Commun. 2025 Aug;46(15):e2500090. doi: 10.1002/marc.202500090. Epub 2025 Apr 14. Macromol Rapid Commun. 2025. PMID: 40229657
-
Aggregation-Induced Emission Boosting the Study of Polymer Science.Macromol Rapid Commun. 2022 Aug;43(16):e2200080. doi: 10.1002/marc.202200080. Epub 2022 Apr 1. Macromol Rapid Commun. 2022. PMID: 35320607 Review.
-
The Marriage of Aggregation-Induced Emission with Polymer Science.Macromol Rapid Commun. 2019 Jan;40(1):e1800568. doi: 10.1002/marc.201800568. Epub 2018 Sep 25. Macromol Rapid Commun. 2019. PMID: 30252992 Review.
Cited by
-
Exploring chiral and achiral properties of novel multilayer 3D polymers: synthesis and characterization.RSC Adv. 2025 Jan 31;15(5):3227-3236. doi: 10.1039/d5ra00233h. eCollection 2025 Jan 29. RSC Adv. 2025. PMID: 39896428 Free PMC article.
-
Synthesis and Characterization of Multilayer 3D Chiral Polymers with Enhanced Optical Properties.Molecules. 2025 Mar 31;30(7):1567. doi: 10.3390/molecules30071567. Molecules. 2025. PMID: 40286155 Free PMC article.
References
-
- Liu Y., Wu G., Yang Z., Rouh H., Katakam N., Ahmed S., Unruh D., Cui Z., Lischka H., Li G. Multi-Layer 3D Chirality: New Synthesis, AIE and Computational Studies. Sci. China Chem. 2020;63:692–698. doi: 10.1007/s11426-019-9711-x. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

