Glycine-Based [3+2] Cycloaddition for the Synthesis of Pyrrolidine-Containing Polycyclic Compounds
- PMID: 39683885
- PMCID: PMC11643519
- DOI: 10.3390/molecules29235726
Glycine-Based [3+2] Cycloaddition for the Synthesis of Pyrrolidine-Containing Polycyclic Compounds
Abstract
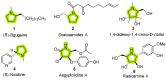
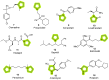
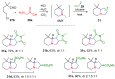
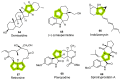
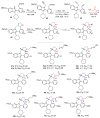
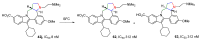
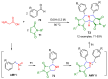
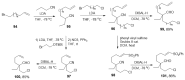
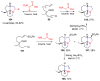
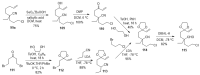
The synthesis of pyrrolidine compounds with biological interest is an active research topic. Glycine could be a versatile starting material for making pyrrolidine derivatives. This review covers recent works on glycine-based [3+2] cycloaddition and combines other annulation reactions in the one-pot synthesis of pyrrolidine-containing heterocyclic compounds. Synthetic method development, substrate scope, and reaction mechanisms are discussed. Applications of the compounds in drug discovery are briefly mentioned. This paper is helpful for chemists in the development of efficient and sustainable methods for the preparation of bioactive pyrrolidine compounds.
Keywords: Glycine; annulation; azomethine ylides; cycloaddition; dipolarophile; pyrrolidine.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



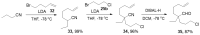
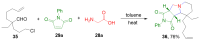
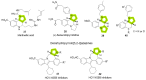
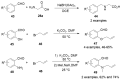
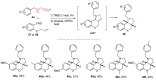


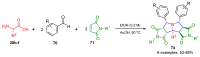
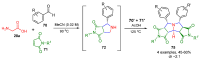

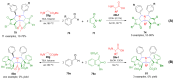
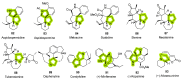
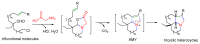

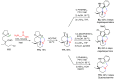

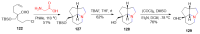
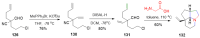
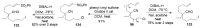


References
Publication types
LinkOut - more resources
Full Text Sources

