Enhancing the Potential of PHAs in Tissue Engineering Applications: A Review of Chemical Modification Methods
- PMID: 39685265
- PMCID: PMC11642177
- DOI: 10.3390/ma17235829
Enhancing the Potential of PHAs in Tissue Engineering Applications: A Review of Chemical Modification Methods
Abstract
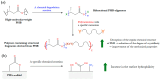
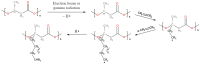
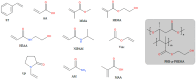
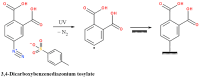
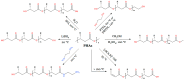
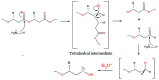
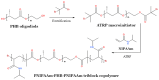
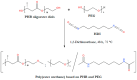
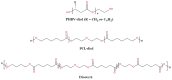
Polyhydroxyalkanoates (PHAs) are a family of polyesters produced by many microbial species. These naturally occurring polymers are widely used in tissue engineering because of their in vivo degradability and excellent biocompatibility. The best studied among them is poly(3-hydroxybutyrate) (PHB) and its copolymer with 3-hydroxyvaleric acid (PHBV). Despite their superior properties, PHB and PHBV suffer from high crystallinity, poor mechanical properties, a slow resorption rate, and inherent hydrophobicity. Not only are PHB and PHBV hydrophobic, but almost all members of the PHA family struggle because of this characteristic. One can overcome the limitations of microbial polyesters by modifying their bulk or surface chemical composition. Therefore, researchers have put much effort into developing methods for the chemical modification of PHAs. This paper explores a rarely addressed topic in review articles-chemical methods for modifying the structure of PHB and PHBV to enhance their suitability as biomaterials for tissue engineering applications. Different chemical strategies for improving the wettability and mechanical properties of PHA scaffolds are discussed in this review. The properties of PHAs that are important for their applications in tissue engineering are also discussed.
Keywords: chemical modifications; functionalized oligomers; polyhydroxyalkanoates (PHAs); polymer grafting; surface modifications; tissue engineering.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
Development and Advantages of Biodegradable PHA Polymers Based on Electrospun PHBV Fibers for Tissue Engineering and Other Biomedical Applications.ACS Biomater Sci Eng. 2021 Dec 13;7(12):5339-5362. doi: 10.1021/acsbiomaterials.1c00757. Epub 2021 Oct 14. ACS Biomater Sci Eng. 2021. PMID: 34649426 Free PMC article. Review.
-
Biosynthesis of poly(3-hydroxybutyrateco-3-hydroxy-4-methylvalerate) by Strain Azotobacter chroococcum 7B.Acta Naturae. 2016 Jul-Sep;8(3):77-87. Acta Naturae. 2016. PMID: 27795846 Free PMC article.
-
Piezoelectric 3-D Fibrous Poly(3-hydroxybutyrate)-Based Scaffolds Ultrasound-Mineralized with Calcium Carbonate for Bone Tissue Engineering: Inorganic Phase Formation, Osteoblast Cell Adhesion, and Proliferation.ACS Appl Mater Interfaces. 2019 May 29;11(21):19522-19533. doi: 10.1021/acsami.9b04936. Epub 2019 May 17. ACS Appl Mater Interfaces. 2019. PMID: 31058486
-
Polyhydroxybutyrate (PHB) in nanoparticulate form improves physical and biological performance of scaffolds.Int J Biol Macromol. 2023 May 1;236:123875. doi: 10.1016/j.ijbiomac.2023.123875. Epub 2023 Mar 3. Int J Biol Macromol. 2023. PMID: 36870657
-
Polyhydroxyalkanoates: Properties and chemical modification approaches for their functionalization.Biotechnol Prog. 2018 Jan;34(1):29-41. doi: 10.1002/btpr.2565. Epub 2017 Oct 16. Biotechnol Prog. 2018. PMID: 28960792 Review.
References
-
- Castro-Sowinski S., Burdman S., Matan O., Okon Y. Natural Functions of Bacterial Polyhydroxyalkanoates. In: Chen G.-Q., editor. Plastics from Bacteria: Natural Functions and Applications. Springer; Berlin/Heidelberg, Germany: 2010. pp. 39–61.
-
- Rai R., Roy I. A Handbook of Applied Biopolymer Technology: Synthesis, Degradation and Applications. The Royal Society of Chemistry; London, UK: 2011. Polyhydroxyalkanoates: The Emerging New Green Polymers of Choice; pp. 79–101.
Publication types
LinkOut - more resources
Full Text Sources

