Optimizing extrusion processes and understanding conformational changes in itraconazole amorphous solid dispersions using in-line UV-Vis spectroscopy and QbD principles
- PMID: 39687500
- PMCID: PMC11647160
- DOI: 10.1016/j.ijpx.2024.100308
Optimizing extrusion processes and understanding conformational changes in itraconazole amorphous solid dispersions using in-line UV-Vis spectroscopy and QbD principles
Abstract
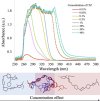
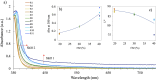
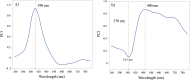
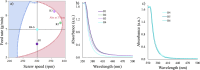
This paper presents a comprehensive investigation of the manufacturing of itraconazole (ITZ) amorphous solid dispersions (ASDs) with Kolllidon® VA64 (KVA64) using hot-melt extrusion (HME) and in-line process monitoring, employing a Quality by Design (QbD) approach. A sequential Design of Experiments (DoE) strategy was utilized to optimize the manufacturing process, with in-line UV-Vis spectroscopy providing real-time monitoring. The first DoE used a fractional factorial screening design to evaluate critical process parameters (CPPs), revealing that ITZ concentration had the most significant impact on the product quality attributes. The second DoE, employing a central composite design, explored the interactions between feed rate and screw speed, using torque and absorbance at 370 nm as responses to develop a design space. Validation studies confirmed process robustness across multiple days, with stable in-line UV-Vis spectra and consistent product quality using 30 % ITZ, 300 rpm, 150 °C and 7 g/min as the optimized process conditions. Theoretical and experimental analyses indicated that shifts in UV-Vis spectra at different ITZ concentrations were due to conformational changes in ITZ, which were confirmed through density functional theory (DFT) calculations and infrared spectroscopy. This work offers novel insights into the production and monitoring of ITZ-KVA64-ASDs, demonstrating that in-line UV-Vis spectroscopy is a powerful tool for real-time process monitoring and/or control.
Keywords: Amorphous Solid Dispersion (ASD); Density Functional Theory (DFT); Design of Experiments (DoE); Hot-Melt Extrusion (HME); Itraconazole (ITZ); Quality by Design (QbD), In-line UV–Vis spectroscopy.
© 2024 The Authors.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures











References
-
- Almeida J., Bezerra M., Markl D., Berghaus A., Borman P., Schlindwein W. Development and validation of an In-Line API quantification method using AQbD Principles based on UV-vis spectroscopy to monitor and optimise continuous hot melt extrusion process. Pharmaceutics. 2020;12(2):150. doi: 10.3390/pharmaceutics12020150. - DOI - PMC - PubMed
-
- Andreas C.J., Rosenberger J., Butler J., Augustijns P., McAllister M., Abrahamsson B., Dressman J. Introduction to the OrBiTo decision tree to select the most appropriate in vitro methodology for release testing of solid oral dosage forms during development. Eur. J. Pharm. Biopharm. 2018;130:207–213. doi: 10.1016/j.ejpb.2018.07.003. - DOI - PubMed
LinkOut - more resources
Full Text Sources

