Relationship Between Body Weight and Leukopenia in Non-Kidney Solid Organ Transplant Recipients With Normal Renal Function Who Are Receiving Valganciclovir for CMV Prophylaxis
- PMID: 39692604
- PMCID: PMC12017309
- DOI: 10.1111/tid.14418
Relationship Between Body Weight and Leukopenia in Non-Kidney Solid Organ Transplant Recipients With Normal Renal Function Who Are Receiving Valganciclovir for CMV Prophylaxis
Abstract
Background: Cytomegalovirus (CMV) disease causes significant morbidity among solid organ transplant (SOT) recipients. To prevent CMV disease, eligible recipients are frequently started on valganciclovir (VGC) prophylaxis post-transplant. Leukopenia has been documented as a primary adverse events of the drug (1). This study's primary aim was to determine whether a patient's weight at the start of VGC prophylaxis was associated with the development of leukopenia.
Methods: This was a single center, retrospective cohort study that included adults > 18 years of age, who had received an organ transplant (heart, liver, or lung) at an academic transplant center from January 1, 2018 through December 31, 2022. A creatinine clearance of > 60 mL/min was required.
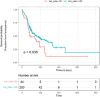
Results: All 294 included patients received 900 mg/day of VGC for CMV prophylaxis, without dose adjustment. Fifty-two percent of the patients developed leukopenia while receiving VGC prophylaxis. The mean weight at initiation of VGC was higher in patients who did not develop leukopenia (97.9 kg) compared to those who did (90.7 kg; p = 0.0112). It was found that with each 1 kg increase in body weight, the likelihood of developing leukopenia decreased by 1.7% (p = 0.004, odds ratio = 0.983, 95% confidence interval [CI], 0.972-0.994). Patients with a baseline body-mass index (BMI) > 25 had a longer median freedom time from leukopenia after initiation of VGC as compared to the group with baseline BMI < 25 (log-rank p = 0.035).
Conclusion: These data suggest that in SOT recipients with normal renal function, receiving a fixed dose of VGC resulted in a significant, inverse relationship between body weight and the development of leukopenia.
Keywords: cytomegalovirus; leukopenia; prophylaxis; transplant; weight.
© 2024 The Author(s). Transplant Infectious Disease published by Wiley Periodicals LLC.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
References
-
- Pappo A., Peled O., Berkovitch M., et al., “Efficacy and Safety of a Weight‐Based Dosing Regimen of Valganciclovir for Cytomegalovirus Prophylaxis in Pediatric Solid‐Organ Transplant Recipients,” Transplantation 103, no. 8 (2019): 1730–1735. - PubMed
-
- Pescovitz M. D., “Valganciclovir,” Transplantation Reviews 20, no. 2 (2006): 82–87.
-
- Paya C., Humar A., Dominguez E. D., et al., “Efficacy and Safety of Valganciclovir Vs. Oral Ganciclovir for Prevention of Cytomegalovirus Disease in Solid Organ Transplant Recipients,” American Journal of Transplantation 4, no. 4 (2004): 611–620. - PubMed
-
- Pescovitz M. D., “Oral Ganciclovir and Pharmacokinetics of Valganciclovir in Liver Transplant Recipients,” Transplant Infectious Disease 1, no. suppl. 1 (1999): 31–34. - PubMed
-
- Fishman J. A., Emery V., Freeman R., et al., “Cytomegalovirus in Transplantation—Challenging the Status Quo,” Clinical Transplantation 21, no. 2 (2007): 149–158. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical