Impact of prolactin treatment on enhancing the cellular responses of MCF7 breast cancer cells to tamoxifen treatment
- PMID: 39692941
- PMCID: PMC11655925
- DOI: 10.1007/s12672-024-01701-x
Impact of prolactin treatment on enhancing the cellular responses of MCF7 breast cancer cells to tamoxifen treatment
Abstract
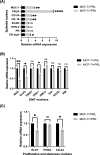
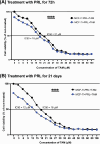
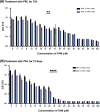
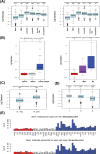
Breast cancer remains one of the most challenging diseases to treat due to its heterogeneity, propensity to recur, capacity to spread to distant vital organs, and, ultimately, patient death. Estrogen receptor-positive illness comprises the most common breast cancer subtype. Preclinical progress is hampered by the scarcity of medication-naïve estrogen receptor-positive tumour models that recapitulate metastatic development and treatment resistance. It is becoming increasingly clear that loss of differentiation and increased cellular stemness and plasticity are important causes of cancer evolution, heterogeneity, recurrence, metastasis, and treatment failure. Therefore, it has been suggested that reprogramming cancer cell differentiation could offer an effective method of reversing cancer through terminal differentiation and maturation. In this context, the hormone prolactin is well recognized for its pivotal involvement in the development of the mammary glands lobuloalveolar tissue and the terminal differentiation that drives the production of the milk protein gene and lactation. Additionally, numerous studies have examined the engagement of prolactin in breast cancer as a differentiation player that resulted in the ablation of tumour growth and progression. Here, we showed that a pre-treatment of the estrogen-positive breast cancer cell line with prolactin led to a considerable improvement in the sensitivity of this cancer cell to Tamoxifen endocrine therapy. We also showed a favourable prognostic value of prolactin receptors/estrogen receptors 1 (or alpha) co-expression on breast cancer patients outcomes, and this co-expression is highly correlated with the well-differentiated breast tumour type. Our results revealed a fruitful aspect of the effects of prolactin in improving the responses of breast cancer cells to conventional endocrine therapy. Moreover, these findings further validated the ability of prolactin as a persuader of a more differentiated and less aggressive breast cancer phenotype. Hence, it suggested a potential implication of prolactin as a therapeutic candidate.
Keywords: Breast cancer; Differentiation; Epithelial markers; Prolactin; Sensitivity; Tamoxifen.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Not applicable. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures


References
-
- Fitzmaurice C, et al. Global, regional, and national cancer incidence, mortality, years of life Lost, years lived with disability, and disability-adjusted life-years for 32 cancer groups, 1990 to 2015: a systematic analysis for the global burden of disease study. JAMA Oncol. 2017;3:524–48. 10.1001/jamaoncol.2016.5688. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
