Future spinal reflex is embedded in primary motor cortex output
- PMID: 39693430
- PMCID: PMC11654683
- DOI: 10.1126/sciadv.adq4194
Future spinal reflex is embedded in primary motor cortex output
Abstract
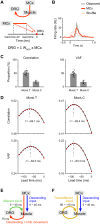
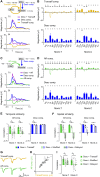
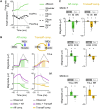
Mammals can execute intended limb movements despite the fact that spinal reflexes involuntarily modulate muscle activity. To generate appropriate muscle activity, the cortical descending motor output must coordinate with spinal reflexes, yet the underlying neural mechanism remains unclear. We simultaneously recorded activities in motor-related cortical areas, afferent neurons, and forelimb muscles of monkeys performing reaching movements. Motor-related cortical areas, predominantly primary motor cortex (M1), encode subsequent afferent activities attributed to forelimb movement. M1 also encodes a subcomponent of muscle activity evoked by these afferent activities, corresponding to spinal reflexes. Furthermore, selective disruption of the afferent pathway specifically reduced this subcomponent of muscle activity, suggesting that M1 output drives muscle activity not only through direct descending pathways but also through the "transafferent" pathway composed of descending plus subsequent spinal reflex pathways. Thus, M1 provides optimal motor output based on an internal forward model that prospectively computes future spinal reflexes.
Figures




References
-
- Felleman D. J., Van Essen D. C., Distributed hierarchical processing in the primate cerebral cortex. Cereb. Cortex 1, 1–47 (1991). - PubMed
-
- Lemon R. N., Descending pathways in motor control. Annu. Rev. Neurosci. 31, 195–218 (2008). - PubMed
-
- Alstermark B., Isa T., Circuits for skilled reaching and grasping. Annu. Rev. Neurosci. 35, 559–578 (2012). - PubMed

