The Epilepsy-Aphasia Syndrome gene, Cnksr2, Plays a Critical Role in the Anterior Cingulate Cortex Mediating Vocal Communication
- PMID: 39694826
- PMCID: PMC11747972
- DOI: 10.1523/ENEURO.0532-24.2024
The Epilepsy-Aphasia Syndrome gene, Cnksr2, Plays a Critical Role in the Anterior Cingulate Cortex Mediating Vocal Communication
Abstract
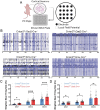
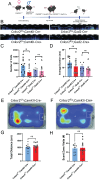
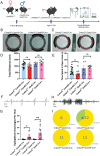
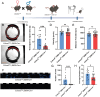
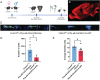
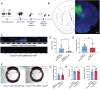
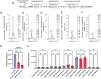
Epilepsy Aphasia Syndrome (EAS) is a spectrum of childhood disorders that exhibit complex co-morbidities that include epilepsy and the emergence of cognitive and language disorders. CNKSR2 is an X-linked gene in which mutations are linked to EAS. We previously demonstrated Cnksr2 knockout (KO) mice model key phenotypes of EAS analogous to those present in clinical patients with mutations in the gene. Cnksr2 KO mice have increased seizures, impaired learning and memory, increased levels of anxiety, and loss of ultrasonic vocalizations (USV). The intricate interplay between these diverse phenotypes at the brain regional and cell type level remains unknown. Here we leverage conditional deletion of the X-linked Cnksr2 in a neuronal cell type manner in male mice to demonstrate that anxiety and impaired USVs track with its loss from excitatory neurons. Finally, we further narrow the essential role of Cnksr2 loss in USV deficits to excitatory neurons of the Anterior Cingulate Cortex (ACC), a region in mice recently implicated in USV production associated with specific emotional states or social contexts, such as mating calls, distress calls, or social bonding signals. Together, our results reveal Cnksr2-based mechanisms that underlie USV impairments that suggest communication impairments can be dissociated from seizures or anxiety. Furthermore, we highlight the cortical circuitry important for initiating USVs.Significance Statement Epilepsy-Aphasia Syndromes are at the severe end of a spectrum of cognitive-behavioral symptoms that are seen in childhood epilepsies and are currently an inadequately understood disorder. The prognosis of EAS is frequently poor and patients have life-long language and cognitive disturbances. We show that the deletion of Cnksr2 specifically within glutamatergic neurons of the anterior cingulate cortex leads to ultrasonic vocalization impairments, providing an important new understanding of the modulation of vocal communication.
Copyright © 2024 Hossain et al.
Conflict of interest statement
The authors declare no competing financial interests.
Figures

References
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
