A General Platform for Copper-Catalyzed Atom Transfer Radical Addition with Electron-Deficient Olefins
- PMID: 39694876
- PMCID: PMC12323560
- DOI: 10.1021/acs.orglett.4c04260
A General Platform for Copper-Catalyzed Atom Transfer Radical Addition with Electron-Deficient Olefins
Abstract
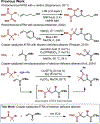
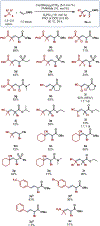
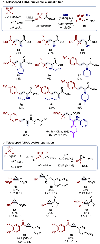
We disclose a broad platform for copper-catalyzed atom transfer radical addition (ATRA) of electron-deficient olefins. Catalytic Cu(dtbbpy)2(OTf)2 enables radical addition of electron-deficient alkyl halides to acrylates, acrylamides, and vinyl sulfones in fair to excellent yields. The resultant ATRA products can be used in a variety of telescoped reactions, including substitution with basic amine nucleophiles to afford α-amino esters. The synthetic utility of the products is further demonstrated through derivatization to give substituted cyclopropanes and a γ,δ-unsaturated ester.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

References
-
- Kharasch MS; Jensen EV; Urry WH Addition of Carbon Tetrachloride and Chloroform to Olefins. Science, 1945, 102, 128. - PubMed
-
- Kharasch MS; Skell PS; Fisher PJ Reactions of Atoms and Free Radicals in Solution. XII. The Addition of Bromo Esters to Olefins. J. Am. Chem. Soc 1948, 70, 1055–1059.
-
- Minisci F Free–Radical Additions to Olefins in the Presence of Redox Systems. Acc. Chem. Res 1975, 8, 165–171.
-
- Williams TM; Stephenson CRJ. Atom Transfer Radical Addition using Photoredox Catalysis. In Visible Light Photocatalysis in Organic Chemistry; Stephenson CRJ; Yoon TP; MacMillan DWC. Wiley: Weinheim, 2018, 73–92.
-
- Engl S; Reiser O Copper-photocatalyzed ATRA Reactions: Concepts, Applications, and Opportunities. Chem. Soc. Rev 2022, 51, 5287–5299. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

