Time-efficient strategies in human iPS cell-derived pancreatic progenitor differentiation and cryopreservation: advancing towards practical applications
- PMID: 39695795
- PMCID: PMC11658428
- DOI: 10.1186/s13287-024-04068-6
Time-efficient strategies in human iPS cell-derived pancreatic progenitor differentiation and cryopreservation: advancing towards practical applications
Abstract
Background: Differentiation of patient-specific induced pluripotent stem cells (iPS) helps researchers to study the individual sensibility to drugs. However, differentiation protocols are time-consuming, and not all tissues have been studied. Few works are available regarding pancreatic exocrine differentiation of iPS cells, and little is known on culturing and cryopreserving these cells.
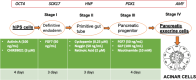
Methods: We differentiated the iPS cells of two pediatric Crohn's disease patients into pancreatic progenitors and exocrine cells, adapting and shortening a protocol for differentiating embryonic stem cells. We analyzed the expression of key genes and proteins of the differentiation process by qPCR and immunofluorescence, respectively. We explored the possibility of keeping differentiated cells in culture and freezing and thawing them to shorten the time needed for the differentiation. We analyzed the cell cycle of undifferentiated and differentiated cells by flow cytometry.
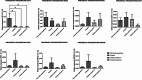
Results: The analysis of mRNA levels of key pancreatic differentiation genes PDX1 and pancreatic amylase indicate that iPS cells were successfully differentiated into pancreatic exocrine cells with expression of PDX1 (one way ANOVA p < 0.0001), and the two isoforms of amylase (one way ANOVA p < 0.05) significantly higher in exocrine cells in comparison to iPS cells. Differentiation efficiency was also confirmed by immunofluorescence analysis of PDX1 and amylase. We confirmed the possibility of shortening the time necessary for obtaining pancreatic cells without losing differentiation efficiency. Pancreatic progenitors and exocrine cells were maintained in culture and cryopreserved. Interestingly, the stemness marker OCT4 resulted significantly lower after subculturing (OCT4 p < 0.001; one-way ANOVA) and after freezing and thawing procedures (p < 0.05, one-way ANOVA) suggesting a reduction of undifferentiated stem cells leading to a purer population of pancreatic progenitor cells. Also, the stemness marker NANOG resulted lower after passaging, corroborating this result.
Conclusions: In this work, we optimized the generation of patient-specific pancreatic differentiated cells and laid the foundation for creating a bank of patient-specific pancreatic lines exploitable for tailored pharmacological assays.
Trial registration: The study was approved by the Ethical Committee of the Institute of Maternal and Child Health IRCCS Burlo Garofolo, with approval number 1556 (internal ID RC 44/22).
Keywords: Crohn’s disease.; Human induced pluripotent stem cells; Pancreatic exocrine cells; Pancreatic progenitors; Patient-specific model.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: The study was approved by the Ethical Committee of the Institute of Maternal and Child Health IRCCS Burlo Garofolo, with approval number 1556 (internal ID of the study RC 07/14 and RC 44/22). Title of the approved project: “ Patient-derived induced pluripotent stem cells for personalizing therapy: the paradigm of thiopurine pancreatitis in Crohn’s disease” and “Comparison of 2D and 3D patient-derived pancreatic exocrine models for the study of thiopurine induced pancreatitis in pediatric IBD patients: an innovative approach therapy personalization”; Name of the institutional approval committee: Ethical Committee of the Institute of Maternal and Child Health IRCCS Burlo Garofolo (Trieste, Italy); Approval number: 1556; Date of approval: 14.07.2014. Informed consent: Obtained from all subjects involved in the study. Competing interests: The authors declare that they have no competing interests.
Figures






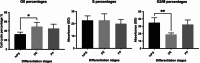
References
-
- Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126(4):663–76. - PubMed
-
- Rispoli P, Scandiuzzi Piovesan T, Decorti G, Stocco G, Lucafò M. iPSCs as a groundbreaking tool for the study of adverse drug reactions: a new avenue for personalized therapy. WIREs Mech Dis. 2024;16(1):e1630. - PubMed
-
- Yamanaka S. A fresh look at iPS cells. Cell. 2009;137(1):13–7. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

