Efficacy and safety of PD-1 and PD-L1 inhibitors in advanced colorectal cancer: a meta-analysis of randomized controlled trials
- PMID: 39696009
- PMCID: PMC11658155
- DOI: 10.1186/s12876-024-03554-8
Efficacy and safety of PD-1 and PD-L1 inhibitors in advanced colorectal cancer: a meta-analysis of randomized controlled trials
Abstract
Background: PD-1 and PD-L1 inhibitors have emerged as promising therapies for advanced colorectal cancer (CRC), but their efficacy and safety profiles require further evaluation. This meta-analysis aims to assess the efficacy and safety of PD-1/PD-L1 inhibitors in this patient population.
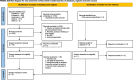
Methods: A systematic review and meta-analysis were conducted following PRISMA guidelines, with data sourced from PubMed, Embase, CENTRAL, Web of Science, and CNKI up to August 3, 2024. Nine randomized controlled trials (RCTs) involving 1680 patients were included. The primary outcomes were overall survival (OS), progression-free survival (PFS) and objective response rate (ORR), while safety was assessed through adverse events (AEs) and grade ≥ 3 AEs. Effect sizes were calculated using mean differences (MD) and risk ratios (RR) with 95% confidence intervals (CIs).
Results: Overall, the meta-analysis showed that PD-1/PD-L1 inhibitors did not significantly extend OS (MD = 0.86, 95% CI: -0.55, 2.27), but they significantly improved PFS (MD = 2.53, 95% CI: 0.92, 4.15). Additionally, PD-1/PD-L1 inhibitors did not significantly increase the ORR compared to controls (RR = 1.19, 95% CI: 0.99, 1.44). In terms of safety, PD-1/PD-L1 inhibitors did not significantly increase the incidence of overall AEs. Subgroup analysis further indicated that PD-1 inhibitors significantly improved OS (MD = 1.24, 95% CI: 0.20, 2.29) and PFS (MD = 6.27, 95% CI: 0.56, 11.97), while PD-L1 inhibitors did not have a significant impact on these outcomes. Additionally, PD-L1 inhibitors were associated with a higher risk of grade ≥ 3 AEs (RR = 1.29, 95% CI: 1.07, 1.57), a risk not observed with PD-1 inhibitors.
Conclusion: PD-1 inhibitors significantly improve PFS and OS in advanced CRC, making them a preferable option over PD-L1 inhibitors, which show limited efficacy and a higher risk of severe AEs. These findings support prioritizing PD-1 inhibitors in clinical practice for this patient group, while caution is warranted with PD-L1 inhibitors due to their safety concerns.
Trial registration: PROSPERO (CRD42024611696).
Keywords: Adverse events; Colorectal cancer; Immunotherapy; Meta-analysis; PD-1; PD-L1.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: This is a systematic review and meta-analysis, ethics approval and consent to participate are not applicable. Consent for publication: Not applicable. The manuscript does not include the participant's identification image or other personal or clinical details. Competing interests: The authors declare no competing interests.
Figures


Similar articles
-
Efficacy and safety of PD-1/PD-L1 inhibitors in advanced or recurrent endometrial cancer: a meta-analysis with trial sequential analysis of randomized controlled trials.Front Immunol. 2025 Jan 31;16:1521362. doi: 10.3389/fimmu.2025.1521362. eCollection 2025. Front Immunol. 2025. PMID: 39958346 Free PMC article.
-
Single or combined immune checkpoint inhibitors compared to first-line platinum-based chemotherapy with or without bevacizumab for people with advanced non-small cell lung cancer.Cochrane Database Syst Rev. 2020 Dec 14;12(12):CD013257. doi: 10.1002/14651858.CD013257.pub2. Cochrane Database Syst Rev. 2020. Update in: Cochrane Database Syst Rev. 2021 Apr 30;4:CD013257. doi: 10.1002/14651858.CD013257.pub3. PMID: 33316104 Free PMC article. Updated.
-
Single or combined immune checkpoint inhibitors compared to first-line platinum-based chemotherapy with or without bevacizumab for people with advanced non-small cell lung cancer.Cochrane Database Syst Rev. 2021 Apr 30;4(4):CD013257. doi: 10.1002/14651858.CD013257.pub3. Cochrane Database Syst Rev. 2021. PMID: 33930176 Free PMC article.
-
Efficacy and safety of PD-1 inhibitors as second-line treatment for advanced squamous esophageal cancer: a systematic review and network meta-analysis with a focus on PD-L1 expression levels.Front Immunol. 2025 Jan 23;15:1510145. doi: 10.3389/fimmu.2024.1510145. eCollection 2024. Front Immunol. 2025. PMID: 39916953 Free PMC article.
-
Efficacy and safety of first-line PD-1/PD-L1 inhibitor in combination with CTLA-4 inhibitor in the treatment of patients with advanced non-small cell lung cancer: a systemic review and meta-analysis.Front Immunol. 2025 Feb 6;16:1515027. doi: 10.3389/fimmu.2025.1515027. eCollection 2025. Front Immunol. 2025. PMID: 39981238 Free PMC article.
Cited by
-
Beta-glucans in oncology: revolutionizing treatment with immune power & tumor targeting.Naunyn Schmiedebergs Arch Pharmacol. 2025 Aug 27. doi: 10.1007/s00210-025-04519-8. Online ahead of print. Naunyn Schmiedebergs Arch Pharmacol. 2025. PMID: 40864254 Review.
References
-
- Morgan E, Arnold M, Gini A, Lorenzoni V, Cabasag CJ, Laversanne M, et al. Global burden of colorectal cancer in 2020 and 2040: incidence and mortality estimates from GLOBOCAN. Gut. 2023;72(2):338–44. - PubMed
-
- Shinji S, Yamada T, Matsuda A, Sonoda H, Ohta R, Iwai T, et al. Recent advances in the treatment of colorectal cancer: a review. J Nippon Med Sch = Nippon Ika Daigaku zasshi. 2022;89(3):246–54. - PubMed
-
- Haller DG. Update on chemotherapy for advanced colorectal cancer. Oncology (Williston Park). 2001;15(3 Suppl 5):11–5. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

