Heterogeneous immune landscapes and macrophage dynamics in primary and lung metastatic adenoid cystic carcinoma of the head and neck
- PMID: 39697346
- PMCID: PMC11653016
- DOI: 10.3389/fimmu.2024.1483887
Heterogeneous immune landscapes and macrophage dynamics in primary and lung metastatic adenoid cystic carcinoma of the head and neck
Abstract
Introduction: Recurrent or metastatic adenoid cystic carcinoma (ACC) of the head and neck is rare and highly aggressive. Due to the ineffectiveness of immune checkpoint therapies, this study aims to investigate the tumor immune microenvironment of primary tumor tissues and lung metastatic tissues and to comprehend the challenges of immunotherapy.
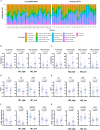
Methods: We analyzed RNA sequencing data and constructed immune landscapes from 25 primary tumors and 34 lung metastases. The data were then validated by immunohistochemistry and single-cell sequencing analysis.
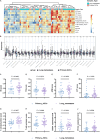
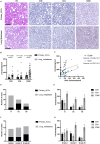
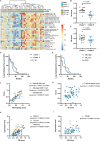
Results: Compared to adjacent normal tissues, both primary and lung metastatic ACC showed low immune infiltration. Lung metastases had higher immune infiltration levels and antigen presentation scores but also higher T cell exclusion and dysfunction scores. Single-cell sequencing data and immunohistochemistry revealed abundant immunosuppressive tumor-associated macrophages in lung metastases. Patients with high M2 macrophage infiltration had shorter lung metastasis-free survival.
Discussion: Primary and lung metastatic ACC exhibit heterogeneous tumor immune microenvironments. Higher immune cell infiltration in lung metastases is countered by the presence of suppressive tumor-associated macrophages, which may limit effective anti-tumor responses.
Keywords: adenoid cystic carcinoma; immune evasion; lung metastasis; tumor immune microenvironment; tumor-associated macrophage.
Copyright © 2024 Wang, Ma, Liu, Zhang, Yang, Zhao, Kong, Gao and Chen.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Figures


References
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

