A Randomized Study to Compare the Efficacy Between Intranasal and Intravenous Dexmedetomidine for the Removal of Foreign Bodies in the Esophagus at the Cricopharynx Level in Pediatric Patients
- PMID: 39697930
- PMCID: PMC11653978
- DOI: 10.7759/cureus.73909
A Randomized Study to Compare the Efficacy Between Intranasal and Intravenous Dexmedetomidine for the Removal of Foreign Bodies in the Esophagus at the Cricopharynx Level in Pediatric Patients
Abstract
Background: Foreign body (coins, magnets, button batteries, and metallic foreign bodies) ingestion is common and causes significant morbidity and mortality in children aged six months to three years. Endoscopic removal of swallowed foreign substances is widely accepted, but sedation and general anesthesia may be required to alleviate pain and anxiety during the procedure. Dexmedetomidine is used as a sedative, hypnotic, anxiolytic, and analgesic. In this study, we aimed to investigate and compare the efficacy between the intranasal (IN) and intravenous (IV) routes for the administration of dexmedetomidine as an adjunct to propofol for the removal of foreign bodies in the esophagus at the cricopharynx level in pediatric patients.
Materials and method: This prospective, double-blinded, randomized study was conducted on 80 patients of either sex and American Society of Anaesthesiologists physical status (ASA PS) I and II aged one month to 12 years with a foreign body in the esophagus at the cricopharynx level undergoing endoscopic foreign body removal. Patients were randomly allocated into groups A and B, with 40 patients each. In the preoperative area group A, IN, 3 mcg/kg, was dripped equally in both nostrils (supine position). In Group B, IV, 3 mcg/kg, diluted in normal saline, was administered intravenously slowly over 10 minutes. The onset of sedation was assessed using the Ramsay Sedation Scale (RSS) every 10 minutes following dexmedetomidine administration, and when an RSS score of 3 or above was achieved, the patient was transferred to the operation theater. In the operation room, patients were induced with injection propofol in a dose of 2 mg/kg or until the loss of verbal response. Parent separation scale scoring was used to evaluate the child's behavior on separation from parents as a tool to assess the anxiolytic property of dexmedetomidine. Perioperative pain using the Wong-Baker Faces pain rating scale, heart rate (HR), oxygen saturation (SpO2), ECG, and blood pressure (BP) were monitored before drug delivery till the end of the procedure. Statistical analysis was done using SPSS version 24.0 (IBM Corp., Armonk, NY).
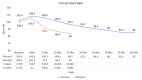
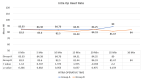
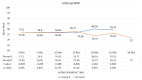
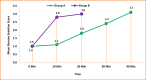
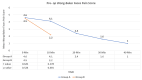
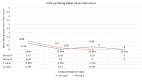
Results: The mean time (minutes) in shifting to operation theater (33.8 ± 5) was significantly higher in group A compared to group B (10.7 ± 2.1) (p = 0.001). The mean Ramsay Sedation Score was higher at 10 and 20 minutes in group B as compared to group A. Group B had significantly lower pain (2.2 ± 1.4) (p ≤ 0.001) at 10 minutes, while in group A, the pain score decreased slowly and took more time but remains lower (1.35 ± 0.94) for longer duration (120 minutes) in the postoperative period in comparison to Group B (1.94 ± 0.35) (p = 0.002). Group B had a sharp hemodynamic decrease, while group A had slow, persistent changes.
Conclusion: IN administration of dexmedetomidine is a safer and more effective mode of sedation to remove a foreign body from the esophagus at the cricopharyngeal level in pediatric patients. It provides stable and sustained hemodynamic parameters and longer postoperative pain relief compared to IV dexmedetomidine.
Keywords: anesthesia; dexmedetomidine; foreign body ingestion; intranasal dexmedetomidine; intravenous dexmedetomidine; pediatric foreign body ingestion; pharyngeal foreign body.
Copyright © 2024, Kumar et al.
Conflict of interest statement
Human subjects: Consent for treatment and open access publication was obtained or waived by all participants in this study. Uttar Pradesh University of Medical Sciences, Saifai, Etawah issued approval 45/2022-23 (494/UPUMS/DSW/Ethical/2022-23). The study was registered at the Clinical Trial Registry of India with Reg. No. CTRI/2023/10/058515. Animal subjects: All authors have confirmed that this study did not involve animal subjects or tissue. Conflicts of interest: In compliance with the ICMJE uniform disclosure form, all authors declare the following: Payment/services info: All authors have declared that no financial support was received from any organization for the submitted work. Financial relationships: All authors have declared that they have no financial relationships at present or within the previous three years with any organizations that might have an interest in the submitted work. Other relationships: All authors have declared that there are no other relationships or activities that could appear to have influenced the submitted work.
Figures




Similar articles
-
Comparison of dexmedetomidine and midazolam for monitored anesthesia care combined with tramadol via patient-controlled analgesia in endoscopic nasal surgery: A prospective, randomized, double-blind, clinical study.Curr Ther Res Clin Exp. 2007 Mar;68(2):69-81. doi: 10.1016/j.curtheres.2007.04.001. Curr Ther Res Clin Exp. 2007. PMID: 24678121 Free PMC article.
-
Comparison of the Effects of Intravenous Fentanyl and Intravenous Dexmedetomidine on Characteristics of Spinal Anesthesia.Cureus. 2024 Oct 24;16(10):e72263. doi: 10.7759/cureus.72263. eCollection 2024 Oct. Cureus. 2024. PMID: 39583493 Free PMC article.
-
Effects of Intranasal and Intravenous Dexmedetomidine on Hemodynamic Responses to Tracheal Intubation and Skull Pin Holder Fixation: A Double-Blinded, Randomized Controlled Trial.Cureus. 2025 Jan 5;17(1):e76980. doi: 10.7759/cureus.76980. eCollection 2025 Jan. Cureus. 2025. PMID: 39912047 Free PMC article.
-
Efficacy of premedication with intranasal dexmedetomidine for removal of inhaled foreign bodies in children by flexible fiberoptic bronchoscopy: a randomized, double-blind, placebo-controlled clinical trial.BMC Anesthesiol. 2019 Dec 2;19(1):219. doi: 10.1186/s12871-019-0892-6. BMC Anesthesiol. 2019. PMID: 31791239 Free PMC article. Clinical Trial.
-
Nebulized Versus Intravenously Administered Dexmedetomidine for Obtunding Hemodynamic Responses to Laryngoscopy and Tracheal Intubation: A Randomized Double-Blind Comparative Study.Cureus. 2024 Feb 23;16(2):e54768. doi: 10.7759/cureus.54768. eCollection 2024 Feb. Cureus. 2024. PMID: 38524064 Free PMC article.
References
-
- Inhaled foreign bodies in children. Brown TC, Clark CM. https://pubmed.ncbi.nlm.nih.gov/6621465/ Med J Aust. 1983;2:322–326. - PubMed
-
- Spontaneous passage of gastrointestinal foreign bodies in children. Lee JH, Nam SH, Lee JH, et al. Pediatr Gastroenterol Hepatol Nutr. 2007;10:157–165.
-
- Respiratory manifestations of the ingestion of a foreign body in pediatric population. Sihem M, Zoheirkermezli Zoheirkermezli. Int J Sci Res. 2021;10:1172–1173.
LinkOut - more resources
Full Text Sources
