Phase II Trial of Neoadjuvant Docetaxel/Cisplatin/5-Fluorouracil Combined with Pegteograstim for Unresectable, Locally Advanced Sinonasal Squamous Cell Carcinoma: KCSG HN18-07
- PMID: 39701091
- PMCID: PMC12263236
- DOI: 10.4143/crt.2024.1025
Phase II Trial of Neoadjuvant Docetaxel/Cisplatin/5-Fluorouracil Combined with Pegteograstim for Unresectable, Locally Advanced Sinonasal Squamous Cell Carcinoma: KCSG HN18-07
Abstract
Purpose: The role of neoadjuvant chemotherapy in locally advanced sinonasal squamous cell carcinoma (SNSCC) has not been established prospectively. We conducted a phase II trial of neoadjuvant chemotherapy (NAC) with docetaxel/cisplatin/5-fluorouracil (TPF) in this population.
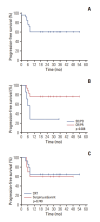
Materials and methods: Eligible patients had unresectable, locally advanced SNSCC, defined as T3/4 category or potential compromise of critical organ function on surgery. Three TPF (docetaxel 75 mg/m2 and cisplatin 75 mg/m2 on day 1, 5-fluorouracil 1,000 mg/m2 on days 1-4 every 3 weeks) cycles were administered with prophylactic pegteograstim. The primary outcome was the objective response rate (ORR); the secondary outcomes included 2-year progression-free survival (PFS), eyeball preservation rate, and safety.
Results: Among 28 patients screened, 25 were evaluable for efficacy (one screen-failure; two evaluable for safety only). The confirmed ORR was 72.0%. The definitive post-NAC treatment comprised chemoradiotherapy (n=15) and surgery (n=10). With a median follow-up of 25.5 months, median PFS was not reached and the 2-year PFS rate was 60.4%. Response to NAC was related to prolonged PFS (p=0.038). No patient underwent eyeball exenteration at the data cutoff point. Treatment-related adverse events of grade ≥ 3 were neutropenia (48.1%) including febrile neutropenia (14.8%), followed by acute kidney injury (22.2%), nausea/vomiting (11.1%), anemia (7.4%), thrombocytopenia (7.4%), and enterocolitis (3.7%).
Conclusion: TPF NAC showed a promising efficacy and might help preserve critical structures in this population, which needs to be validated in a large prospective trial (KCT0003377).
Keywords: Nasal cavity; Neoadjuvant chemotherapy; Paranasal sinus; Prognosiss; Squamous cell carcinoma.
Conflict of interest statement
Pegteograstim was provided by Green Cross Cooperation (Korea).
Figures


Similar articles
-
A prospective phase II randomized study of docetaxel combined with lobaplatin versus TPF regimen induction chemotherapy followed by concurrent chemoradiotherapy for locally advanced head and neck squamous cell carcinoma.J Cancer Res Clin Oncol. 2023 Dec;149(20):18081-18091. doi: 10.1007/s00432-023-05497-1. Epub 2023 Nov 20. J Cancer Res Clin Oncol. 2023. PMID: 37985501 Free PMC article. Clinical Trial.
-
Phase II Trial of Induction Chemotherapy for Advanced Sinonasal Squamous Cell Carcinoma.Clin Cancer Res. 2025 Jan 17;31(2):258-265. doi: 10.1158/1078-0432.CCR-24-1416. Clin Cancer Res. 2025. PMID: 39531535 Clinical Trial.
-
Taxane, platinum and 5-FU prior to chemoradiotherapy benefits patients with stage IV neck node-positive head and neck cancer and a good performance status.J Cancer Res Clin Oncol. 2018 Feb;144(2):389-401. doi: 10.1007/s00432-017-2553-9. Epub 2017 Dec 8. J Cancer Res Clin Oncol. 2018. PMID: 29222650 Free PMC article.
-
Gemcitabine-based chemotherapy for advanced biliary tract carcinomas.Cochrane Database Syst Rev. 2018 Apr 6;4(4):CD011746. doi: 10.1002/14651858.CD011746.pub2. Cochrane Database Syst Rev. 2018. PMID: 29624208 Free PMC article.
-
Chemotherapy for advanced gastric cancer.Cochrane Database Syst Rev. 2017 Aug 29;8(8):CD004064. doi: 10.1002/14651858.CD004064.pub4. Cochrane Database Syst Rev. 2017. PMID: 28850174 Free PMC article.
References
-
- Ackall FY, Issa K, Barak I, Teitelbaum J, Jang DW, Jung SH, et al. Survival outcomes in sinonasal poorly differentiated squamous cell carcinoma. Laryngoscope. 2021;131:E1040–8. - PubMed
-
- Dutta R, Dubal PM, Svider PF, Liu JK, Baredes S, Eloy JA. Sinonasal malignancies: a population-based analysis of site-specific incidence and survival. Laryngoscope. 2015;125:2491–7. - PubMed
-
- Lee MM, Vokes EE, Rosen A, Witt ME, Weichselbaum RR, Haraf DJ. Multimodality therapy in advanced paranasal sinus carcinoma: superior long-term results. Cancer J Sci Am. 1999;5:219–23. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

