Establishing an immune correlate of protection for Nipah virus in nonhuman primates
- PMID: 39702562
- PMCID: PMC11659318
- DOI: 10.1038/s41541-024-01036-2
Establishing an immune correlate of protection for Nipah virus in nonhuman primates
Abstract
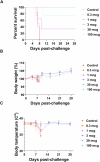
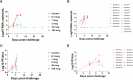
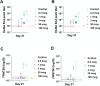
The limited but recurrent outbreaks of the zoonotic Nipah virus (NiV) infection in humans, its high fatality rate, and the potential virus transmission from human to human make NiV a concerning threat with pandemic potential. There are no licensed vaccines to prevent infection and disease. A recombinant Hendra virus soluble G glycoprotein vaccine (HeV-sG-V) candidate was recently tested in a Phase I clinical trial. Because NiV outbreaks are sporadic, and with a few cases, licensing will likely require an alternate regulatory licensing pathway. Therefore, determining a reliable vaccine correlate of protection (CoP) will be critical. We assessed the immune responses elicited by HeV-sG-V in African Green monkeys and its relationship with protection from a NiV challenge. Data revealed values of specific binding and neutralizing antibody titers that predicted survival and allowed us to establish a mechanistic CoP for NiV Bangladesh and Malaysia strains.
© 2024. The Author(s).
Conflict of interest statement
Competing interests: V.H.L.G., D.P., G.G.L., and S.H. are Auro Vaccines LLC employees. C.C.B. is a US federal employee and co-inventor on US and foreign patents pertaining to soluble forms of the Hendra virus and Nipah virus G glycoproteins whose assignee is the United States as represented by the Henry M. Jackson Foundation for the Advancement of Military Medicine. Soluble forms of the Hendra virus and Nipah virus G glycoproteins are licensed to Zoetis Inc. and Auro Vaccines LLC. The remaining authors declare no competing interests.
Figures
References
-
- Moore, K. A. et al. Measures to prevent and treat Nipah virus disease: research priorities for 2024-29. Lancet Infect Dis. Published online July 2024. 10.1016/S1473-3099(24)00262-7 (2024). - PubMed
Grants and funding
- U19AI142764/U.S. Department of Health & Human Services | National Institutes of Health (NIH)
- UC7AI094660/U.S. Department of Health & Human Services | National Institutes of Health (NIH)
- U19AI142764/U.S. Department of Health & Human Services | NIH | Office of Extramural Research, National Institutes of Health (OER)
- UC7AI094660/U.S. Department of Health & Human Services | NIH | Office of Extramural Research, National Institutes of Health (OER)
LinkOut - more resources
Full Text Sources

